Abstract
Purpose of Review
Intensive forest management practices are being implemented worldwide to meet future global demand for wood and wood products while facilitating the protection of natural forest ecosystems. A potential decline in wood properties associated with rapid tree growth makes it essential to quantify the potential impact of intensive management on the process of wood formation and, in turn, on its suitability for various end-uses.
Recent Findings
Wood produced over short rotations is generally of lower quality because wood properties tend to improve with cambial age (i.e. the number of annual growth rings from the pith). The intensification of silvicultural practices can thus have measurable consequences for the forest products value chain. The use of new planting material from tree improvement programs could offset such effects, but questions arise as to the effects of a changing climate on wood produced from these plantations and the best silvicultural approaches to manage them.
Summary
Based on these recent findings, we provide reflections on the need for a modelling framework that uses the effects of cambial age, ring width and position along the stem to summarise the effects of tree growth scenarios on wood properties. We then present challenges related to our limited understanding of the effects of several drivers of wood properties, such as climate variation, genetic material, and forest disturbances, among others, and highlight the need for further data collection efforts to better anticipate the quality attributes of the future wood fibre resource. We conclude by providing examples of promising new tools and technologies that will help move wood quality research forward by allowing (1) fast, efficient characterisation of wood properties, and (2) up-scaling predictions at the landscape level to inform forest management decisions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Commonly understood definitions of intensive forest management practices have been in use for over 70 years [1], with adjustments made over subsequent decades by specialists in various sub-disciplines. However, most definitions in use today [2–7] converge around the principle that silvicultural treatments can be used to maximise wood and timber production. These silvicultural treatments include plantations of hybrid or fast-growing tree species (e.g. [8]), respacing (e.g. [9]), thinning (e.g. [10]), pruning (e.g. [11]), fertilisation (e.g. [12]), and vegetation control (e.g. [13])—with or without herbicides, insecticides or biocontrol of pests (e.g. [14]).
Currently, the most commonly mentioned solution in the scientific literature to meet future global demand for wood while allowing the protection of natural forest ecosystems relates to increasing wood supply from plantations [2, 7]. Several jurisdictions around the world integrate the use of planted forests into their strategy for forest conservation [15–20]. As the adoption of intensive forest management practices increases, it is essential to consider their potential impact on the process of wood formation and, in turn, on wood quality. Indeed, silvicultural practices often result in important changes in growth patterns and tree form, which can lead to variability in wood properties, thereby influencing suitability for various end-uses [21, 22].
Pioneering work from Philip R. Larson in the 1960s [23] described the physiological processes underlying the deleterious effects of rapid growth on wood properties and emphasised that the concept of wood quality must consider specific end-uses. According to this principle—highlighted by many authors in recent years—the definition of wood quality may vary according to the desired end-product [24–28]. For the timber industry, for example, criteria such as wood density, relative proportions of heartwood and sapwood, proportion of juvenile wood (also referred to as corewood) and reaction wood, knots (size, status, frequency, etc.) and grain orientation are the most important to consider, while for the pulp and paper industries, fibre length, wood cell properties and chemical composition are the most relevant [29].
To better anticipate the consequences of silvicultural choices on future wood supplies, there is a need to understand the drivers of variation in wood properties [2, 25]. In turn, such knowledge can help increase supply chain productivity and improve economic performance.
The intensification of silvicultural practices has measurable consequences for the forest products value chain. In the USA, for example, serious concerns were raised in 2010 regarding the bending design values for southern pine lumber from planted forests, which were found to be significantly lower than those obtained from natural forests. Consequently, the Southern Pine Inspection Bureau issued new reduced design values for some grades of southern pine dimension lumber [30]. This allowed the construction industry to use lumber from planted forests under revised building codes [31]. Such adjustments further highlight the relative nature of the wood quality concept; not only does it vary according to different end-uses, but it can also evolve over time for a specific product.
With this reality in mind, we aimed to assemble current knowledge on the effects of intensive silvicultural practices on important wood properties. This way, readers can interpret the information provided based on their own understanding of wood quality. Our review builds upon previous review papers on the effect of silvicultural treatments on wood properties [25–29, 32–34]. Here, we focus more specifically on the effects of intensive silviculture on wood properties with an emphasis on coniferous species from around the world.
In line with current definitions of intensive forest management, papers considered in this review investigated the effects of ‘initial spacing’ thinning, respacing, pruning, fertilisation, site fertility, or genetics on wood properties. We used Google Scholar as our chosen search engine, as a preliminary comparison had shown it was the most inclusive among similar tools. The search terms included the following keywords: ‘the effect(s) of’, plus ‘initial spacing’, ‘thinning’, ‘respacing’, or ‘pruning’, ‘fertilization’ or ‘site fertility’ or ‘genetic(s)’ plus ‘wood properties’, ‘mechanical properties’, ‘wood density’, ‘lumber’, or ‘microfibril angle’, plus ‘plantation’. Once the search was completed, titles and abstracts of highlighted papers were sifted, and only relevant studies were included in the review. Main results from the retained papers were then summarised in the form of tables or graphs to facilitate the analysis. Figure 1 shows the location of each study included in this review.
The review is divided into four main sections. The first section summarises current knowledge on the effects of intensive forest management practices on wood properties. It is based on the main research findings extracted from the relevant empirical studies retained from our literature search. The subsequent sections provide reflections on (i) current knowledge gaps and future challenges, (ii) the need for a modelling framework to summarise the effects of intensive management practices on future wood properties under a changing climate and (iii) the emerging technologies in wood characterisation and utilisation that will help move wood quality research forward. We conclude by highlighting the detrimental effects of a decreasing age at harvest on wood quality and its implications for future production and processing.
Current Knowledge on the Effects of Intensive Forest Management Practices on Wood Properties
Initial Spacing
The choice of initial spacing at planting has an important effect on the secondary growth of trees, wood properties and volume production. Traditionally, the most common spacing used for conifer species was set at approximately 2 m in an orthogonal grid, which is equivalent to ~ 2500 seedlings per hectare [34–36]. However, in recent decades planting distances have tended to increase to encourage faster radial growth, leading to shorter rotations [34, 37–40] and reduction in establishment costs [41, 42]. In New Zealand, for example, typical stocking rates are now around 1,000 seedling per hectare, which represents an average square spacing of 3.2 m [43].
However, trees planted and grown at wider spacings generally have larger crowns that produce a larger juvenile core [32, 34, 39, 44, 45], which contains wood with lower density, higher MFA [46, 47] (Fig. 2, Table 1), larger knots [34, 37, 39, 48–50], greater stem taper [34, 51], increased stem curvature [52], and poor stem straightness in the bottom log [47, 52]. The greater presence of sweep and leaning stems in a stand have also been associated with increased production of compression wood [34], which is associated with high longitudinal shrinkage and a greater propensity to sudden failure under loads than normal wood [53]. These changes in tree morphology and wood properties may eventually alter sawing patterns and thus affect sawmill recovery and efficiency [47, 54–56].
Wider spacings are generally associated with lower modulus of elasticity (MOE) and modulus of rupture (MOR) in coniferous species [37, 46–48, 50, 55, 57] (Fig. 2, Table 1). The effect of wide spacing on wood density, however, is often less severe and varies among species. For example, lower wood density was associated with wider spacings in jack pine (Pinus banksiana Lamb.) [58], Scots pine (Pinus sylvestris L.), Norway spruce (Picea abies (L.) Karst.) [57] and patula pine (Pinus patula Schiede ex Schltdl. et Cham.) [47]. Other studies revealed only slight reductions in wood density at wide spacings in Norway spruce [59], black spruce (Picea mariana (Mill) B.S.P.) [50], and white spruce (Picea glauca (Moench) Voss) [37]. Finally, wood density did not vary significantly with spacing in Sitka spruce (Picea sitshensis (Bong.) Carr) [60], radiata pine (Pinus radiata D. Don) [46], Japanese cedar (Cryptomeria japonica) [48], young jack pine [39] and western hemlock (Tsuga heterophylla (Raf.) Sarg) [61] (Fig. 2, Table 1). As can be seen from the jack pine studies [39, 58], these effects also vary according to tree age.
Most research findings indicate a decrease in fibre length [44, 46, 58, 61] with increased spacing (Fig. 2, Table 1), with the exception of white spruce [45]. A reduction in cell wall thickness at wider spacings has also been reported in radiata pine [46].
While the adverse effects on wood quality attributes of planting at wider spacing are well documented, on some occasions this practice may also be beneficial. For example, on low fertility sites, or under serious drought conditions, wider spacing between trees can be advantageous, as more nutrients and water could be available to individual trees [62].
Thinning and Respacing
Thinning and respacing are intermediate treatments used to favour vigorous, healthy trees, which most likely will increase in size due to accelerated radial growth, thereby improving economic returns [34, 63]. Respacing (or pre-commercial thinning) is applied to young stands before canopy closure and generally before individual stems have reached merchantable size. The effects of respacing on wood properties are comparable to those of initial spacing [9, 64–67] (Table 2). Commercial thinning, in contrast, is usually applied to older stands after canopy closure, which corresponds, more or less, with the time of death of the lowest branches. Its effects on wood supplies are twofold. First, trees removed during thinning operations may include trees with low vigour and poor stem form, among other defects. Second, thinned trees are often of lower quality because they have not reached maturity and may thus contain a higher proportion of corewood [22]. However, little research has been dedicated to quantifying the specific properties of thinned stems. Instead, most work has focused on the effects of thinning on the properties of the crop at final harvest. Due to their older age and acceleration of radial growth at a later stage, commercial thinning may lead to a lower proportion of corewood in the remaining trees [68], while having little effect on knot size [34, 63].
The timing and intensity of thinning or respacing treatments are thus critical, as implementing them too early or too aggressively may result in lower wood density [9, 69, 70], larger knots [65, 67, 71], inferior mechanical properties [65, 72], shorter fibres [73] and lower grade lumber [10, 65], all of which have been reported in trees grown at excessively wide initial spacing (> 2.5 m, Fig. 2).
When thinning treatments are applied optimally, the impact on structural wood quality or chemical composition can either be negligible [74–78], minor [69, 79], or even beneficial [80]. Likewise, the production of compression wood has both been reported to increase [64, 81] or decrease [75] after thinning (Table 2). Results of the latter study were interpreted as a potential response to phototropic stimuli, which might favour the inclination of stems as trees compete for space in unthinned stands. Compared with unthinned stands, trees in thinned stands may have the advantage to exhibit accelerated self-pruning of dead branches, because of the increased effects of wind, rain, snow, and solar radiation inside the stand [82, 83]. In contrast, thinning may contribute to a faster lateral expansion of the tree crown [84]. Thinning has also been reported to stimulate the formation of epicormic branches in Sitka spruce, especially when combined with a high lift pruning treatment [85].
Pruning
Artificial pruning is used to increase the length of the knot-free section of a log, which can increase both mechanical properties [34, 86, 87] and lumber value [33], especially in the most valuable lowest log of elite trees [32, 88]. Knots cause local grain deviation in wood [89], which can cause reduced wood stiffness and strength [86, 87]. A key effect of artificial pruning is to considerably shorten the time required to complete branch occlusion. This leads to fewer loose knots (i.e. knots formed after branch death). For example, the occlusion time for Norway spruce branch stubs can reach 50 years or more under self-pruning conditions [90], while it can occur after 4 to 12 years in pruned trees (2-cm-thick branch wounds) [89].
Usually, artificial pruning of branches is implemented on the lowest branches at an early age, i.e. when they are small, healthy and covered by a thin layer of bark [91]. Early intervention is recommended to minimise the problems of infections and to facilitate rapid occlusion [32, 91]. However, pruned trees can be more susceptible to damage from browsing, or bark stripping and fraying, than unpruned trees [89]. Consequently, the density of ungulate populations should be considered before applying pruning treatments [89, 92].
Other than damage from wildlife, several other factors may influence the response of trees to pruning. These include timing (i.e. seasonality), site fertility, soil moisture availability, climate, tree age, species, the tools used and pruning height [93, 94]. According to Zobel [32], the optimum time for pruning is when the tree has reached about 12 cm in diameter at breast height. While Zobel [32] recommended removal of 33% of the live crown, more recent studies have shown it is possible to remove 50% of the live crown without a significant reduction in radial growth [11, 95, 96] or height growth [95]. However, lift pruning height has also been associated with the development of numerous epicormic sprouts, which can vary importantly according to species [85]. As highlighted in the same study, the relevance of using such a treatment should be considered carefully as pruning is a very expensive treatment that could end up costing two to three times the cost of thinning a stand. When applied, the frequency and severity of pruning treatments should be planned to allow for crown recovery and to limit the effects on growth, which can alter the dominance of pruned trees in the stand [93, 94]. In some cases, tree pruning residues are considered as a useful feedstock for bioenergy, which can help mitigate dependence on fossil fuels [97, 98].
Since pruning artificially increases crown base height, it is generally assumed that wood within the pruned area will reach mature characteristics earlier than in unpruned trees [11, 32, 64]. This implies that microfibril angle (MFA) would decrease while fibre length and wood density would slightly increase after pruning [64]. However, recent studies from Chiu et al. [77] in Taiwania (Taiwania Cryptomerioides Hay) and in Douglas-fir (Pseudotsuga menziesii (Mirb.) Franco) [28, 99] did not find any effects of pruning on MFA, tracheid length, or wood density. In addition, some authors have suggested that heartwood formation can be regulated by pruning [100, 101], although others disagree with this finding [102].
Pruning is often done in combination with thinning [71, 77, 89, 96] or fertilising treatments [95, 103]. Pruning can help limit the increase in stem taper generally caused by thinning [93], although whether this effect can be observed depends on stand density and age [94]. Pruning can also be applied to dead branches. Unlike pruning of live branches, pruning dead branches does not disrupt tree growth [94]. Its only effect would be to reduce the occurrence of loose knots.
Fertilisation
Fertilisation is often portrayed as a temporary increase in site fertility [104]. It can be used to help seedling establishment, especially on nutrient-poor sites. It can also accelerate growth and help tree development during the period of rapid juvenile growth (when nutrient demand is high), albeit to the detriment of wood quality. In the USA, mid-rotation fertilisation is a proven method for improving tree growth by counteracting nutrient limitations [12, 105]. Even late-rotation fertilisation 5 to 10 years before harvesting has been practiced to improve tree growth of Douglas fir in Canada [106]. The area of forest treated with fertilisation has thus increased substantially in recent years [105]. Most often, the applied macronutrients are nitrogen, phosphorus and potassium (NPK). Their effects on wood quality depend on several factors, including the combination of nutrients applied, their concentration, the preexisting site conditions [107] and the species being treated [108].
Most studies measuring the effects of fertilisation treatments on wood properties have focused on wood density, with variable responses reported (Table 3). Some studies indicated a reduction in wood density in conifers after fertilisation [105, 106, 109–111], while others found either no decrease in density or only a slight decrease [74, 112, 113]. The effect on wood density also tends to last for a limited period of time, i.e. around 5 years following treatment (e.g. [109, 111]). Generally, the reduction in wood density is more pronounced when fertilisation is applied at higher concentrations. For example, Antony et al. [105] showed a reduction in wood density of about 40 kg m−3 in a thinned loblolly pine (Pinus taeda L.) plantation after a mid-rotation application of heavy nitrogen fertiliser treatment (336 kg ha−1), compared with the unfertilised control. The response of wood density to fertilisation treatments may also depend on the additional effects of stand density management operations.
Often, fertilisation is applied in combination with thinning treatments. For example, a recent study by Kantavichai et al. [114] reported that the decline in wood density following fertilisation was dependent on both initial stocking and the intensity of pre-commercial thinning treatments in Douglas-fir plantations in the US Pacific Northwest. According to their study, trees that produced the highest density and stiffest wood, came from stands that had been pre-commercially thinned to half their original stem density without further thinning and fertilisation treatments. Usually, when fertilisation and thinning are combined, the radial growth response is greater than when only fertilisation treatments are applied [112, 113].
Aside from wood density, few studies have examined the effects of fertilisation on other wood properties. As for wood density, the effects on wood fibre characteristics seem to be more pronounced when heavy fertilisation treatments are applied. For example, Antony et al. [105] found a significant decrease in cell wall thickness and a significant increase in tracheid radial diameter under a heavy fertilisation treatment (336 kg ha−1 of N). At lower concentrations (e.g. 112 kg ha−1 or 224 kg ha−1 of N), the effects on wood fibre characteristics were either not significant or negligible. Mäkinen and Hynyen [112] found only small differences between the fertilisation treatments (control vs fertilised with 150 kg N + 75 kg P2O5 + 75 kg K2O ha−1) in the earlywood: latewood ratio (3–9%), tracheid diameter (2–5%), tracheid length (4–5%) and cell wall thickness (0–10%) in Scots pine. Small increases in MFA after fertilisation have been reported, which led to a slight decrease in stiffness in loblolly pine and radiata pine [105, 110]. To reduce the negative impacts of fertilisation on wood mechanical properties, Downes et al. [110] suggested applying mild fertilisation treatments more frequently. In terms of chemical properties, nitrogen fertilisation has been reported to increase lignin and extractive concentrations [107, 115], which could affect pulp bleaching processes in pulp and paper manufacturing.
Site
Silviculturists usually quantify site productivity using site index, which is typically defined as the average height of the dominant trees in even-aged stands at a given index age [116]. Higher values are therefore associated with higher overall site productivity. Since site conditions influences tree growth, it is generally assumed that wood properties will be affected too [24]. Studies looking at the link between site index and wood properties often show a decrease in wood properties with an increase in site index. For example, Antony et al. [117] observed that both MOE and MOR values decrease in loblolly pine with an increase in site index. Their observations were consistent with earlier ones made by Brazier and Mobbs [60] in Sitka spruce. Watson and Bradley [118] also reported a decrease in wood density with an increase in site index in several commercial conifer species, while fibre length showed the opposite trend.
Silviculturists can also use the concept of site quality to characterise a site. In this case, site quality is assessed using physical and biological variables—such as water and nutrient availability, temperature and precipitation, latitude or soil type—or using a classification system that stratifies the landscape into ecologically meaningful units [116]. Generally, most studies on the effects of site quality on wood properties have focused on wood density; however, recent efforts have been made to predict site level or regional variation in MOE and MOR for some coniferous species (e.g. [117, 119]). For example, it has been reported that the MOE and MOR values of loblolly pine and radiata pine increased on warmer sites [117, 119]. It is believed that air temperature measured in the autumn is the most important and consistent variable influencing MOE values as it influences the production of latewood [120]. As such, sites benefiting from an extending growing season were also found to result in lower MFA [121].
Temperature, rainfall and site fertility have also been recognised as environmental drivers of wood density (e.g. [122]). For example, in radiata pine growing in New Zealand, higher wood density was observed in warmer sites at lower elevations [122]. Moore et al. and Rossi et al. [65, 123] also demonstrated that wood density decreased with increasing elevation and latitude in Sitka spruce and black spruce, respectively, while Giroud et al. [124] showed no effect of a latitudinal gradient on wood density in black spruce, jack pine and balsam fir. In black spruce, wood density was also found to be higher on either very dry (e.g. [125]) or wet sites (e.g. [126]).
Our review of literature has shown that studies based on site index tend to report contrasting results to those including site quality and climatic variables. It is thus difficult to summarise the effect of site productivity or site quality on wood properties. To better understand the variation in wood properties along a gradient of site conditions or site productivity, many different variables or combinations of variables should be considered and tested. Often, the influence of site productivity is studied in combination with climatic conditions and genetic factors (e.g. [127]). To meet the challenge of adapting forest management practices to climate change impacts, it will also be important for future studies to disentangle the effects of site from those of genotype on key determinants of wood quality [125].
Genetics
Genetic improvement of forest trees, or tree breeding programs, have long been considered important for increasing growth rates and wood quality with respect to certain traits of interest, such as wood density. However, as plantation rotations were reduced over time, foresters and tree breeders realised that fast-grown trees contained a large proportion of corewood with unfavourable wood properties. This motivated forest geneticists to include more wood quality traits in tree improvement programs [128–131]. Traits such as wood density, microfibril angle, fibre properties, stiffness and dimensional stability are now commonly included in tree breeding programs. Among the traits tested, wood density, acoustic velocity (used to estimate wood stiffness), and microfibril angle appear to be the most heritable (Table 4). High narrow-sense heritability (i.e. the proportion of phenotypic variance due to additive genetic variation) values and their low standard errors suggest high genetic control for these traits [129]. However, estimates of individual narrow-sense heritability often vary with cambial age (i.e. the number of annual growth rings from the pith) and among species, which complicates the task of genetic selection [132–134].
Depending on the wood quality traits evaluated, heritability was found to increase with cambial age [133–135], while in some cases, it reached maximum values at a specific cambial age [133, 136]. In addition, some traits of interest, such as wood stiffness, are highly correlated with other traits like microfibril angle and wood density, which themselves are not always well correlated (Table 5). Correlations between wood quality traits may also be complicated by environmental factors (genotype-by-environment interactions) [136–138]. To better control the often negative correlations between tree growth and wood quality characteristics, tree breeders stress the need for improving their understanding of the molecular genetics of wood traits [8, 139].
Current Knowledge Gaps and Future Challenges
Over the past 40 years, the Earth’s temperature has risen by 0.18 °C per decade [140], which has resulted in an average increase in tree growing seasons of approximately 3.6 days per decade in Europe [141]. There are geographical variations in such effects, and in some regions, an extended growing season has already caused measurable phenological changes in trees [142–145], which could lead to changes in wood properties. Despite recent efforts, few studies have conducted in-depth investigations of the effects of climate warming on wood properties. According to a review on the wood anatomy of boreal species under a changing climate by Zhang et al. [26], the proportion of latewood in coniferous species appears to be negatively correlated with climate warming, while the proportion of earlywood and fibre length would likely increase with warming temperatures.
It is anticipated that water availability will be the most important limiting factor to tree growth in the twenty-first century, thus exceeding temperature across large portions of the boreal zone [146•]. In areas, where acute drought stress is expected to increase, such as in Central Europe and western North America, changes in wood cell formation might be more pronounced [62, 147]. This may affect conifers, as tracheid length is largely driven by the water availability during cell formation [148, 149]. For example, Jyske et al. [69] observed an increase in the cell wall thickness of both earlywood and latewood tracheids of Norway spruce following induced drought. Despite MFA being considered to be under strong genetic control, a decrease in MFA has been observed under drought conditions [26]. Earlywood cell adaptations are believed to support hydraulic functional responses to climate, while latewood cells are believed to support mechanical functional responses to climate [150].
Drought-induced changes such as thicker cell walls and lower MFA may prove beneficial in terms of wood mechanical properties. However, drought can also cause significant reductions in annual radial growth and height increment [147], as well as drastically increasing the risk of tree mortality [151]. Such effects could largely overcome potential benefits in terms of wood properties. In comparison to broadleaved species, conifers have the tendency to maintain larger margins of safety from hydraulic failure, which may favour their chances of survival during drought periods [152]. However, this margin of safety varies between species according to their water potential regulation strategies (i.e. isohydric vs anisohydric) [151].
With the observed and predicted increases in global temperatures and changes in precipitation and radiation budgets, forests will also become more susceptible to a wider range of natural disturbances. While many uncertainties remain regarding changes in disturbance dynamics under climate change [153–155], warmer and drier conditions are expected to increase the frequency and severity of wildfire, drought and insect disturbances, while warmer and wetter conditions are likely to increase disturbances arising from wind and pathogens [154].
It remains uncertain how forest management practices should be adapted in response to climate change and new forest disturbance regimes [156]. Intensive forest management scenarios with short rotation lengths may bear the key advantage of reducing exposure time, thereby increasing the likelihood of a given crop reaching the final harvest prior to being disturbed. From a wood supply perspective, shorter rotations can confer an additional advantage of reduced risk of loss from catastrophic disturbances because younger stands tend to be less vulnerable to various hazards such as wind damage (e.g. [157]), insect attacks (e.g. [158]), drought stress (e.g. [159]) and, to some extent, wildfire (e.g. [160]). Following drought events, Bennett et al. [159] found that larger trees tend to suffer from higher mortality rates, which also suggests that shorter rotations can lead to a reduced risk. However, Büntgen et al. [161] reported that fast-grown trees may be more vulnerable to biotic stressors.
To reduce risk at the landscape level, one suggestion is to reduce stand density through more intensive thinning to limit growth restrictions caused by drought (e.g. [62]) or to increase resistance to insect attacks (e.g. [162–164]). In their systematic review of the effects of thinning, Moreau et al. [165•] revealed a generally positive influence of thinning on forest resistance and resilience to stressors such as fire, drought, insects and pathogens. However, multiple factors may influence these processes, and the review, as well as other studies [157, 166], also highlighted a tendency for windthrow resistance to decrease temporarily after thinning.
Despite uncertainties about future wind regimes, increases in wind exposure may also alter wood cell development. Under high wind exposure, conifers are more likely to develop compression wood [167], which is characterised by higher wood density, higher microfibril angle, shorter fibre or tracheid length, higher proportion of lignin, and thicker cell walls [22] than normal mature wood. The high exposure time needed to induce a compression wood response [167] suggests a significant increase in sustained wind speeds would be required to directly affect wood quality. However, if the occurrence of extreme wind events causes root systems to tilt rather than fully overturning (e.g. [168]), this will inevitably lead to the production of compression wood in conifers. This is a concern because the presence of this type of wood in solid wood products can lead to sudden failure under load stresses [34, 53, 169].
Genetic improvement programs have started to investigate the impact of different biotic and abiotic agents on wood quality traits, but several questions remain. For example, Nabais et al. [125] recently questioned the assumed link between wood density and drought tolerance and highlighted that, for some species, other variables, such as tree competition, soil fertility or resistance to pathogens, are important drivers of wood density variation that may undermine the assumed density-drought relationship. Therefore, they suggested that selection for drought tolerance should not systematically be based on wood density as a proxy. Other studies testing the relationships between tree resistance to insects and diseases on the one hand, and tree growth or wood quality traits on the other, are providing new knowledge that could guide tree improvement strategies. For example, Lenz et al. [170] recently showed that weevil resistance in Norway spruce is genetically positively correlated with other traits such as tree height, height-to-diameter ratio, and wood acoustic velocity. It is still unclear, however, how these relationships will vary among species and which wood quality traits might serve as reliable proxies of tree resistance to insects and diseases [171]. An important challenge to this work is the time required to confirm the efficacy and durability of the resistance of various genotypes to diseases or insects in different environments. A proposed way to overcome this challenge is to combine breeding with genetic engineering [172].
In summary, a key challenge of future wood quality research will be to predict the impact of intensive management practices under changing climatic conditions and a higher frequency and intensity of disturbances from biotic or abiotic stressors. Recent literature provides clear evidence that trees adapt their wood cell development in response to variations in temperature, water availability and CO2 atmospheric concentration. However, linking environmental conditions to wood properties remains an emerging field of research. Further studies will be required before we can fully predict the effects of climate change—and its interactions with silvicultural treatments—on future wood quality attributes. An important limitation of the empirical approach generally used in wood quality studies is that the applicability of results is generally restricted to the range of tested conditions. It is practically impossible to test each potential combination of species, site, genetic material, silvicultural scenarios, and climatic conditions under which trees are managed. Also, the need to sample mature stands to evaluate the full impacts of silvicultural decisions implies that timely questions can only be answered several decades later (at least in temperate and boreal forests). In the meantime, the genetic material, silvicultural approach, species of interest, and even the wood properties of interest will have likely changed.
The Need for a Modelling Framework
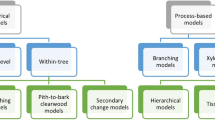
Wood quality models can be used to anticipate wood properties in various conditions that may not have been empirically tested. A review by Drew et al. [173••] summarised recent progress in wood quality modelling and proposed a simple classification in two general groups, i.e. fully empirical and process-based. Here, we focus on the former and highlight how empirical knowledge such as that included in this review can be assembled into a statistical modelling framework that facilitates the extrapolation of results to untested conditions. This approach is based on the work of Larson [23], who proposed that the distinctive radial patterns of wood physico-mechanical properties were largely the result of crown processes, such that the type of xylem formed depends on both the proximity to the live crown and the age of the cambial initials, due to the differential distribution of growth regulators (i.e. auxin gradient) along the stem. Further understanding of the process of xylem formation in trees and of developmental constraints on cambial maturation led to the formulation of competing, but overlapping, hypotheses that the distinctive radial patterns of wood properties is driven by the need for optimising both mechanical stability and hydraulic efficiency in tree stems [174]. These concepts led to the development of a stem quality modelling approach whereby the systematic within-stem variation in wood properties is described as functions of cambial age, height in the stem and annual ring width, the latter being used as a proxy for crown vigour [175–179]. This modelling strategy allowed early assessments and comparisons of the impacts of any silvicultural scenarios on wood properties provided that their effects on tree growth could be simulated, i.e. predictions from growth models were used as input to the wood properties models [180].
Because this statistical modelling approach has the capacity to address the effects of multiple, potentially confounding factors [181], it has been widely used to detect and quantify the impact of drivers of wood properties variation, including site characteristics [126], silviculture [182] and genetics [183]. However, the use of annual ring width (or ring area) as a proxy for the effects of environmental conditions has important limitations, as both silvicultural treatments [47, 182] and climatic conditions [184, 185] have been shown to influence wood properties beyond the effects that could be anticipated from their effects on tree growth.
To improve model predictions, climatic variables are increasingly included as predictors of wood properties variation in such models (e.g. [70, 186–188]). One approach is to develop correlations or response functions between seasonal or monthly climatic variables on the one hand, and annual ring-level wood properties on the other [186], but this may hinder the possibility of detecting the effects of acute climatic events such as frost and droughts that can alter wood properties [189, 190]. Dendroclimatological approaches have been developed to quantify the effects of acute climatic events on tree growth [191, 192], which could be applied to studies of wood properties. Babst et al. [193] provide a comprehensive synthesis of models developed to simulate radial growth as a function to climatic variables from dendrochronological measurements. Models driven by temperature, water balance and day length could offer a promising approach for simulating wood properties, which could be used in the described framework.
Key to informing silvicultural decision-making is the integration of wood properties models into growth modelling platforms. Ideally, these platforms should include risk assessment modules and allow simulations of changes in forest disturbance regimes under different climate projections [2, 25, 194, 195]. Not only should platforms be user-friendly, but they should also follow the free and open source (FOSS) philosophy, so that the information is widely accessible, and the workflow is transparent and fully reproducible. CAPSIS is one notable example of such a platform that has been used extensively and has fostered international collaborations [196]. In addition to informing silvicultural decision-making before empirical tests can yield results, the developed platforms provide an identifiable locus for the integration of the most up-to-date knowledge on the links between growth conditions and wood properties. Such integration is key to meeting the challenges associated with ensuring that both wood supply and wood quality from intensively managed forests continues to meet demands associated with new product development.
Emerging Technologies in Wood Characterisation and Utilisation
New technologies offer fresh opportunities for fast and efficient characterisation of wood properties. For example, recent efforts have led to new algorithms or models for assessment of wood density and mechanical properties through high-resolution scanning technologies such as computed tomography (CT) (e.g. [197, 198], micro-CT [199], and other methods using a combination of X-ray densitometry and ultrasonic measurements [200]. As they offer the possibility to characterise wood properties rapidly and at a fine scale, such technologies will facilitate greater knowledge acquisition on the effects of intensive forest management practices on wood quality.
A concomitant challenge is the need to expand wood properties assessments to scales that are relevant to the strategic decisions that must be made in forest management. Airborne laser scanning (ALS) data has been used to link statistical descriptors of forest inventory metrics to wood properties measured on sample plots, to produce wall-to-wall, landscape-level assessments of wood properties [201–203]. As they have yet to include explicit links between ALS metrics, stand structure and wood properties, such models require local parameterisation and validation. The fusion of terrestrial laser scanning (TLS) and ALS data may help include such explicit links between canopy structure, tree crown characteristics and wood properties, and thus improve model precision transferability across multiple scales [204]. Further development could include the use of spectral information from satellite imagery, which can complement the characterisation of forest structure given by ALS data by providing longitudinal estimates of photosynthetic activity or vegetation stress [205, 206].
At the other end of the forest value chain, it will also be crucial to work closely with the engineered wood products industry to understand which wood quality traits most closely determine the suitability of wood for use in specific products [129, 207]. The detrimental effects of intensive forest management practices on wood traits may be at least partially offset by innovations in wood processing technology. For example, corewood of fast-grown conifers can be used for the central portion of laminated beams or its fibre added to concrete or plastic composites [2]. Other examples include the production of oriented strand board (OSB), particleboard, and medium density fibreboard [208••]. Moore and Cown [208••] argued that corewood panels would have similar mechanical properties and internal bond strength to those from outerwood panels, although their dimensional stability could be compromised. Different processing options should be explored to develop wood products that provide the best possible alignment between market demand and the properties of the resource [209].
Conclusion
All things considered, the most important effects of intensive silvicultural practices on wood properties are related to the trend of decreasing age at harvest (i.e. rotation age) [25, 32, 34, 174, 208••, 210]. With advances in genetic improvement, trees can more rapidly reach a merchantable size, which leads to shorter rotations. This, in turn, leads to higher proportions of corewood, which is characterised by lower mechanical properties, poorer dimensional stability and overall lower quality of lumber for most end-uses [33, 34].
As highlighted in this review, cambial age is a prime determinant of the within-stem variation in wood properties [174]. Many physico-mechanical properties, such as wood density, and strength and stiffness, are low near the pith, increase rapidly in the first few growth rings of the juvenile wood period and then attain more stable values at higher cambial ages [175–177, 211]. Some properties, such as longitudinal shrinkage and microfibril angle (MFA), follow a decreasing trend with cambial age [211–213], and in these cases, lower values are generally more desirable. Some coniferous species have high density near the pith, but this is generally associated with a high microfibril angle, and thus has properties analogous to that of compression wood (also referred to as ‘flexure’ wood) [22, 214].
Overall, the increasing demand for wood fibre combined with the needs to manage the risk of disturbance and protect natural forest ecosystems will imply that an increasing proportion of timber will come from intensively managed plantations at the global scale. Our review has highlighted that genetic selection can help mitigate the impact of a shift towards intensive forest management on future wood quality. However, the importance of cambial age as a driver of the variation of wood properties implies that for most species, sites, genetic material and end-uses, shorter rotations will come at the expense of overall wood quality.
In intensive silvicultural scenarios, the magnitude of this detrimental effect will obviously depend on the chosen rotation length, among other factors; however, it will also depend on the distribution of annual rings within the stem, which can be modified by the application of thinning, fertilising or pruning treatments. Delayed thinning, pruning and delayed harvesting can all help increase the proportion of mature wood in timber. Precise recommendations of silvicultural scenarios that can strike a balance between the needs to produce timber rapidly and maintain adequate wood properties will need to be devised for each species, genetic material and site of interest. Key to achieving this is the development of wood properties models and their integration into growth modelling simulators. Such simulation platforms exist but are only available for a limited number of species and sites. Although they offer an ideal framework to consider the effects of future climate on wood properties, this knowledge has rarely been included in wood quality simulators.
As several jurisdictions make the transition from visual grading of solid wood products to non-destructive mechanical grading [215], there is a growing concern that the lumber produced from intensively managed stands may not always meet the requirements and the grading specifications of the end-users. As highlighted in this review, the wood processing industry can adapt to some extent to the supply of wood of lesser quality, but some thresholds should not be exceeded. The minimum requirement of the C16 mechanical grade for timber frame construction in Europe provides an example of such threshold [65, 216]. A better understanding of the links between silvicultural practices, visual grading, wood properties, and non-destructive mechanical grading is thus required [2, 217]. This highlights the importance for silviculturists, wood scientists and tree breeders to work collaboratively to ensure that future wood quality will not deteriorate under rapidly changing conditions. A desired outcome of such collaborative efforts is that practitioners are given access to the knowledge and material that will allow them to select species and provenances with the best resistance and resilience to stressors without sacrificing either wood volume or quality.
Data Availability
No new data were generated or analysed in support of this research.
References
Papers of particular interest, published recently, have been highlighted as: • of importance •• of major importance
Chapman HH. Forest management. Bristol, Connecticute: The Hildreth Press; 1950.
Gardiner B, Moore J. Creating the wood supply of the future. In: Fenning T, editor. Challenges and opportunities for the world’s forests in the 21st century. Springer; 2014. pp. 677–704.
Tittler R, Filotas É, Kroese J, Messier C. Maximizing conservation and production with intensive forest management: it’s all about location. Environ Manage. 2015;56(5):1104–17. https://doi.org/10.1007/s00267-015-0556-3.
Paquette A, Messier C. The role of plantations in managing the world’s forests in the Anthropocene. Front Ecol Environ. 2010;8(1):27–34. https://doi.org/10.1890/080116.
Bell FW, Pitt DG, Wester MC. Is intensive forest management a misnomer? An Ontario-based discussion of terminology and an alternative approac. For Chron. 2006;82(5):662–74. https://doi.org/10.5558/tfc82662-5.
Bell FW, Parton J, Stocker N, Joyce D, Reid D, Wester M, et al. Developing a silvicultural framework and definitions for use in forest management planning and practice. For Chron. 2008;84(5):678–93. https://doi.org/10.5558/tfc84678-5.
Park A, Wilson ER. Beautiful Plantations: can intensive silviculture help Canada to fulfill ecological and timber production objectives? For Chron. 2007;83(6):825–39. https://doi.org/10.5558/tfc83825-6.
Wu H, Ivkovic M, Gapare W, Matheson A, Baltunis B, Powell M, et al. Breeding for wood quality and profit in Pinus radiata: a review of genetic parameter estimates and implications for breeding and deployment. NZ J Forest Sci. 2008;38(1):56–87.
Auty D, Moore J, Achim A, Lyon A, Mochan S, Gardiner B. Effects of early respacing on the density and microfibril angle of Sitka spruce wood. Forestry: An Int J Forest Res. 2017;91(3):307–19. https://doi.org/10.1093/forestry/cpx004.
Krajnc L, Farrelly N, Harte AM. The effect of thinning on mechanical properties of Douglas fir, Norway spruce, and Sitka spruce. Ann For Sci. 2019;76(1):3.
Gartner BL, Robbins JM, Newton M. Effects of pruning on wood density and tracheid length in young Douglas-fir. Wood Fiber Sci. 2005;37(2):304–13.
Blazier MA, Scott DA, Coleman R. Mid-rotation silviculture timing influences nitrogen mineralization of loblolly pine plantations in the mid-south USA. Forests. 2015;6(4):1061–82.
Thiffault N, Roy V. Living without herbicides in Québec (Canada): historical context, current strategy, research and challenges in forest vegetation management. Eur J Forest Res. 2011;130(1):117–33.
Rolando C, Baillie B, Withers T, Bulman L, Garrett L. Pesticide use in planted forests in New Zealand. N Z J For. 2016;61(2):3–10.
Binkley CS. Preserving nature through intensive plantation forestry: the case for forestland allocation with illustrations from British Columbia. For Chron. 1997;73(5):553–9. https://doi.org/10.5558/tfc73553-5.
Hartmann H, Daoust G, Bigué B. Negative or positive effects of plantation and intensive forestry on biodiversity: A matter of scale and perspective. For Chron. 2010;86(3):354–64. https://doi.org/10.5558/tfc86354-3.
Ferraz SFB, Lima WdP, Rodrigues CB. Managing forest plantation landscapes for water conservation. Forest Ecol Manag. 2013;301:58–66. https://doi.org/10.1016/j.foreco.2012.10.015.
Barrette M, Leblanc M, Thiffault N, Paquette A, Lavoie L, Bélanger L, et al. Issues and solutions for intensive plantation silviculture in a context of ecosystem management. For Chron. 2014;90(06):748–62. https://doi.org/10.5558/tfc2014-147.
Sedjo RA, Botkin D. Using foret plantations to spare natural forests. Environ: Sci Policy Sustain Dev. 1997;39(10):14–30.
Gladstone WT, Thomas LF. Reducing pressure on natural forests through high-yield forestry. For Ecol Manage. 1990;35(1):69–78. https://doi.org/10.1016/0378-1127(90)90232-Z.
Zobel BJ, van Buijtenen JP. Wood variation and wood properties. In: Zobel BJ, van Buijtenen JP, editors. Wood variation: its causes and controls. Springer, Springer Berlin, Heidelberg; 1989. pp. 1–32.
Panshin AJ, De Zeeuw C. Textbook of wood technology: structure, identification, properties, and uses of the commercial woods of the United States and Canada. 4th ed. New York, NY, (USA): McGraw-Hill Book Company; 1980.
Larson PR. Wood formation and the concept of wood quality. Bulletin no 74 New Haven, CT: Yale University, School of Forestry; 1969. p. 1–54. https://elischolar.library.yale.edu/cgi/viewcontent.cgi?article=1085&context=yale_fes_bulletin.
Jozsa L, Middleton G. A discussion of wood quality attributes and their practical implications. Special Publication No. SP-34. Forintek Canada Corp. Vancouver, BC; 1994.
Moore J, Cown D. Wood quality variability—what is it, what are the consequences and what we can do about it. N Z J For. 2015;59:3–9.
Zhang S, Belien E, Ren H, Rossi S, Huang J. Wood anatomy of boreal species in a warming world: a review. iForest – Biogeosci Forestry. 2020;13(2):130–8. https://doi.org/10.3832/ifor3230-013.
Haygreen JG, Bowyer JL. Forest products and wood science: an introduction. 3rd ed. Ames Iowa (USA); Iowa state university press; 1996.
Briggs DG, Smith R. Effects of silvicultural practices on wood properties of conifers: a review. In Oliver C, Hanley D, Johnson J editors. Douglas fir: stand management for the future. Seattle: University of Washington Press; 1986. pp. 108–117.
Listyanto T, Nichols JD. A review of relationships between wood quality and silvicultural practices. Jurnal Ilmu Kehutanan. 2009;3(2):116–26.
SPIB. Determination of design values for visually graded southern pine dimensional lumber. In: Bureau SPI, editor. Penascola, Fl. Lumber Grading Rules | SPIB | Southern; 2012. Pine Inspection Bureau. https://www.spib.org/wood-services/lumber-grading-rules.
Wood Design Focus. A Journal of contemporary wood engineering: 2012 Building Code Updates. Forest Products Society. 2012;22(4)
Zobel B. Silvicultural effects on wood properties. IPEF International. 1992;2(6):31–8.
Moore JR. Growing fit-for-purpose structural timber: what’s the target and how do we get there. N Z J For. 2012;57(3):17–24.
Macdonald E, Hubert J. A review of the effects of silviculture on timber quality of Sitka spruce. Forestry. 2002;75(2):107–38.
Liziniewicz M, Ekö PM, Agestam E. Effect of spacing on 23-year-old lodgepole pine (Pinus contorta Dougl. Var. latifolia) in southern Sweden. Scand J For Res. 2012;27(4):361–71.
Barrette J, Giroud G, Ward C, Serban L, Barrette M. Synthèse des connaissances sur la densité initiale de reboisement. Ministère des Forêts de la Faune et des Parcs: Direction de la recherche forestière. AT-SSRF-19. 2020. p. 25.
Duchesne I, Tanguay F. Effects of initial spacing on tree and wood attributes on lumber quality and value in white spruce - the stanley spacing trial case study: FPInnovations Report. 2011. p. 34.
Groot A, Cortini F. Effects of initial planting density on tree and stand development of planted black spruce up to age 30. For Chron. 2016;92(02):200–10. https://doi.org/10.5558/tfc2016-039.
Hébert F, Krause C, Plourde P-Y, Achim A, Prégent G, Ménétrier J. Effect of tree spacing on tree level volume growth, morphology, and wood properties in a 25-year-old Pinus banksiana plantation in the boreal forest of Quebec. Forests. 2016;7(11):276.
McClain KM, Morris DM, Hills SC, Buse LJ. The effects of initial spacing on growth and crown development for planted northern conifers: 37-year results. For Chron. 1994;70(2):174–82. https://doi.org/10.5558/tfc70174-2.
Huang C-H, Kronrad GD, Morton JD. The financially optimal loblolly pine planting density and management regime for nonindustrial private forestland in East Texas. South J Appl For. 2005;29(1):16–21.
Zhang S, Lei Y, Bowling C. Quantifying stem quality characteristics in relation to initial spacing and modeling their relationship with tree characteristics in black spruce (Picea mariana). North J Appl For. 2005;22(2):85–93.
NZFFA. NZFFA guide sheet no. 1: an introduction to growing radiata pine. 2007. https://www.nzffa.org.nz/farm-forestry-model/resource-centre/information-leaflets/nzffa-guide-sheets-2007/nzffa-guide-sheet-no-1/. Accessed 2021.
Yang K, Hazenberg G. Impact of spacing on tracheid length, relative density, and growth rate of juvenile wood and mature wood in Picea mariana. Can J For Res. 1994;24(5):996–1007.
Yang K-C. Impact of spacing on juvenile wood and mature wood properties of white spruce (Picea glauca). Taiwan J For Sci. 2002;17(1):13–29.
Lasserre J-P, Mason EG, Watt MS, Moore JR. Influence of initial planting spacing and genotype on microfibril angle, wood density, fibre properties and modulus of elasticity in Pinus radiata D. Don corewood. For Ecol Manag. 2009;258(9):1924–31.
Erasmus J, Kunneke A, Drew DM, Wessels CB. The effect of planting spacing on Pinus patula stem straightness, microfibril angle and wood density. Forestry: Int J For Res. 2018;91(3):247–58. https://doi.org/10.1093/forestry/cpy005.
Ishiguri F, Kasai S, Yokota S, Iizuka K, Yoshizawa N. Wood quality of sugi (Cryptomeria japonica) grown at four initial spacings. IAWA J. 2005;26(3):375–86.
Hein S, Weiskittel AR, Kohnle U. Effect of wide spacing on tree growth, branch and sapwood properties of young Douglas-fir [Pseudotsuga menziesii (Mirb.) Franco] in south-western Germany. Eur J For Res. 2008;127(6):481–93.
Zhang S, Chauret G, Ren H, Desjardins R. Impact of plantation black spruce initial spacing on lumber grade yield, bending properties and MSR yield. Wood Fiber Sci. 2002;34(3):460–75.
Smith JHG. Influences of spacing on radial growth and percentage latewood of Douglas-fir, western hemlock, and western redcedar. Can J For Res. 1980;10(2):169–75.
Belley D, Beaudoin M, Duchesne I, Vallerand S, Tong Q, Swift D. Assessment of white spruce and jack pine stem curvature from a Nelder spacing experiment. Wood Fiber Sci. 2013;45(3):237–49.
Sonderegger W, Mandallaz D, Niemz P. An investigation of the influence of selected factors on the properties of spruce wood. Wood Sci Technol. 2008;42(4):281–98.
Belley D, Duchesne I, Vallerand S, Barrette J, Beaudoin M. Computed tomography (CT) scanning of internal log attributes prior to sawing increases lumber value in white spruce (Picea glauca) and jack pine (Pinus banksiana). Can J For Res. 2019;49(12):1516–24. https://doi.org/10.1139/cjfr-2018-0409.
Charlton RA, Naghizadeh Z, Ham C, Wessels CB. A value chain comparison of Pinus patula sawlog management regimes based on different initial planting densities and effect on wood quality. Forest Policy Econ. 2020;111:102067.
Yerbury M, Cooper R. Curve sawing spruce sawlogs containing sweep can reduce drying distortion when compared with conventional sawing. Forestry. 2010;83(4):443–50.
Šilinskas B, Varnagirytė-Kabašinskienė I, Aleinikovas M, Beniušienė L, Aleinikovienė J, Škėma M. Scots Pine and Norway Spruce Wood Properties at Sites with Different Stand Densities. Forests. 2020;11(5):587.
Kang K-Y, Zhang SY, Mansfield SD. The effects of initial spacing on wood density, fibre and pulp properties in jack pine (Pinus banksiana Lamb.). Holzforschung. 2004;58(5):455–63.
Johansson K. Effects of initial spacing on the stem and branch properties and graded quality of Picea abies (L.) Karst. Scand J For Res. 1992;7(1–4):503–14.
Brazier J, Mobbs I. The influence of planting distance on structural wood yields of unthinned Sitka spruce. Forestry: Int J For Res. 1993;66(4):333–52.
Watson P, Garner C, Robertson R, Reath S, Gee W, Hunt K. The effects of initial tree spacing on the fibre properties of plantation-grown coastal western hemlock. Can J For Res. 2003;33(12):2460–8. https://doi.org/10.1139/x03-171.
Rais A, van de Kuilen J-WG, Pretzsch H. Growth reaction patterns of tree height, diameter, and volume of Douglas-fir (Pseudotsuga menziesii [Mirb] Franco) under acute drought stress in Southern Germany. Eur J For Res. 2014;133(6):1043–56.
Moore J. Wood properties and uses of Sitka spruce in Britain. Forestry Commission Research Report, Forestry Commission, Edinburgh. 2011; p. 1–48.
Barbour RJ, Marshall DD, Lowell EC. Managing for wood quality. In: Monserud RA, Haynes RW, Johnson AC, editors. Compatible forest management. Managing Forest Ecosystems, vol 8. Springer. Dordrecht; 2003. pp. 299–336.
Moore J, Achim A, Lyon A, Mochan S, Gardiner B. Effects of early re-spacing on the physical and mechanical properties of Sitka spruce structural timber. For Ecol Manage. 2009;258(7):1174–80.
Watt MS, Zoric B, Kimberley MO, Harrington J. Influence of stocking on radial and longitudinal variation in modulus of elasticity, microfibril angle, and density in a 24-year-old Pinus radiata thinning trial. Can J For Res. 2011;41(7):1422–31.
Auty D, Weiskittel AR, Achim A, Moore JR, Gardiner BA. Influence of early re-spacing on Sitka spruce branch structure. Ann For Sci. 2012;69(1):93–104. https://doi.org/10.1007/s13595-011-0141-8.
Brüchert F, Becker G, Speck T. The mechanics of Norway spruce [Picea abies (L.) Karst]: mechanical properties of standing trees from different thinning regimes. For Ecol Manag. 2000;135(1):45–62. https://doi.org/10.1016/S0378-1127(00)00297-8.
Jyske T, Kaakinen S, Nilsson U, Saranpää P, Vapaavuori E. Effects of timing and intensity of thinning on wood structure and chemistry in Norway spruce. Holzforschung. 2010;64(1):81–91.
Filipescu CN, Lowell EC, Koppenaal R, Mitchell AK. Modeling regional and climatic variation of wood density and ring width in intensively managed Douglas-fir. Can J For Res. 2014;44(3):220–9. https://doi.org/10.1139/cjfr-2013-0275.
Wang S-Y, Lin C-J, Chiu C-M. Effects of thinning and pruning on knots and lumber recovery of Taiwania (Taiwania cryptomerioides) planted in the Lu-Kuei area. J Wood Sci. 2003;49(5):444–9.
Wang X, Ross RJ. Acoustic evaluation of Alaskan young-growth wood. In: Ross RJ, editors. Proceedings of the 15th International Symposium on Nondestructive Testing of Wood: Sept. 10-12, 2007. Madison: Forest Products Society; 2008. pp. 97–105.
Hussein A, Gee W, Watson P, Zhang S. Effect of precommercial thinning on residual sawmill chip kraft pulping and pulp quality in balsam fir. Wood Fiber Sci. 2007;38(1):179–86.
Jaakkola T, Mäkinen H, Saranpää P. Wood density in Norway spruce: changes with thinning intensity and tree age. Can J For Res. 2005;35(7):1767–78.
Cameron A, Thomas K. Effect of thinning on the development of compression wood in stems of Corsican pine. Eur J Forest Res. 2008;127(3):247–51.
Guller B. The effects of thinning treatments on density, MOE, MOR and maximum crushing strength of Pinus brutia Ten. wood. Annals For Sci. 2007;64(4):467–75.
Chiu C-M, Lin C-J, Wang S-Y. Tracheid length and microfibril angle of young Taiwania grown under different thinning and pruning treatments. Wood Fiber Sci. 2007;37(3):437–44.
Pfister O, Wallentin C, Nilsson U, Ekö P-M. Effects of wide spacing and thinning strategies on wood quality in Norway spruce (Picea abies) stands in southern Sweden. Scand J For Res. 2007;22(4):333–43. https://doi.org/10.1080/02827580701504951.
Tong Q, Tanguay F, Zhang S. Impact of commercial thinning on annual radial growth and wood density in plantation-grown black spruce. Wood Fiber Sci. 2011;43(3):311–25.
Russo D, Marziliano PA, Macri G, Proto AR, Zimbalatti G, Lombardi F. Does thinning intensity affect wood quality? An analysis of Calabrian Pine in Southern Italy using a non-destructive acoustic method. Forests. 2019;10(4):303.
Barnett J, Jeronimidis G. editors. Wood quality and its biological basis. CRC Press; 2003.
Mäkinen H. Growth, suppression, death, and self-pruning of branches of Scots pine in southern and central Finland. Can J For Res. 1999;29(5):585–94.
Liziniewicz M, Ekö PM, Klang F. Effects of five tree-selection strategies when thinning spruce (Picea abies) stands: a case study in a field trail in southern Sweden. Scand J For Res. 2016;31(5):495–506.
Ganbaatar B, Jamsran T, Gradel A, Sukhbaatar G. Assessment of the effects of thinnings in scots pine plantations in Mongolia: a comparative analysis of tree growth and crown development based on dominant trees. For Sci Technol. 2021;17(3):135–43. https://doi.org/10.1080/21580103.2021.1963326.
Deal RL, Barbour RJ, Mcclellan MH, Parry DL. Development of epicormic sprouts in Sitka spruce following thinning and pruning in south-east Alaska. Forestry: Int J For Res. 2003;76(4):401–12. https://doi.org/10.1093/forestry/76.4.401.
As N, Goker Y, Dundar T. Effect of Knots on the physical and mechanical properties of scots pine. Wood Res. 2006;51(3):51–8.
Rocha MFV, Costa LR, Costa LJ, Araújo ACCd, Soares BCD, Hein PRG. Wood knots influence the modulus of elasticity and resistance to compression. Wood Science and Technology. Floresta Ambient. 2018;25(4). https://doi.org/10.1590/2179-8087.090617.
Mederski PS, Szczawiński D, Giefing DF, Naparty K, Brunka M. Knot soundness and occlusion time after the artificial pruning of oak. For Res Pap. 2019;80(1):5–11.
Baders E, Donis J, Snepsts G, Adamovics A, Jansons A. Pruning effect on Norway spruce (Picea abies (L.) Karst.) growth and quality. For Stud. 2017;66(1):33–48.
Mäkinen H, Ojansuu R, Sairanen P, Yli-Kojola H. Predicting branch characteristics of Norway spruce (Picea abies (L.) Karst.) from simple stand and tree measurements. Forestry. 2003;76(5):525–46.
O’Hara KL. Pruning wounds and occlusion: a long-standing conundrum in forestry. J Forest. 2007;105(3):131–8.
Hörnberg S. Changes in population density of moose (Alces alces) and damage to forests in Sweden. For Ecol Manage. 2001;149(1–3):141–51.
O’Hara KL. Technical commentary: A biological justification for pruning in coastal Douglas-fir stands. West J Appl For. 1991;6(3):59–63.
Pinkard E, Beadle C. A physiological approach to pruning. Int For Rev. 2000;2(4):295–305.
Amateis RL, Burkhart HE. Growth of young loblolly pine trees following pruning. For Ecol Manage. 2011;262(12):2338–43.
Moreno-Fernández D, Sánchez-González M, Álvarez-González JG, Hevia A, Majada JP, Canellas I, et al. Response to the interaction of thinning and pruning of pine species in Mediterranean mountains. Eur J Forest Res. 2014;133(5):833–43.
Sagani A, Hagidimitriou M, Dedoussis V, editors. Techno-economic evaluation of tree pruning biomass fired power plants for electricity generation: the case of three prefectures in Greece. Proc. 22nd European Biomass Conference and Exhibition; 2014.
Laganière J, Paré D, Thiffault E, Bernier PY. Range and uncertainties in estimating delays in greenhouse gas mitigation potential of forest bioenergy sourced from Canadian forests. Gcb Bioenergy. 2017;9(2):358–69.
Gartner BL. Assessing wood characteristics and wood quality in intensively managed plantations. J Forest. 2005;103(2):75.
Taylor AM, Gartner BL, Morrell JJ. Heartwood formation and natural durability-a review. Wood Fiber Sci. 2002;34(4):587–611.
Margolis HA, Gagnon RR, Pothier D, Pineau M. The adjustment of growth, sapwood area, heartwood area, and sapwood saturated permeability of balsam fir after different intensities of pruning. Can J For Res. 1988;18(6):723–7.
Bergström B, Gref R, Ericsson A. Effects of pruning on heartwood formation in Scots pine trees. J For Sci. 2004;50(1):11–6.
Wiseman D, Smethurst P, Pinkard L, Wardlaw T, Beadle C, Hall M, et al. Pruning and fertiliser effects on branch size and decay in two Eucalyptus nitens plantations. For Ecol Manage. 2006;225(1–3):123–33.
Saarsalmi A, Mälkönen E. Forest fertilization research in Finland: a literature review. Scand J For Res. 2001;16(6):514–35.
Antony F, Jordan L, Schimleck LR, Daniels RF, Clark A III. The effect of mid-rotation fertilization on the wood properties of loblolly pine (Pinus taeda). IAWA J. 2009;30(1):49–58.
Filipescu CN, Trofymow JA, Koppenaal RS. Late-rotation nitrogen fertilization of Douglas-fir: growth response and fibre properties. Can J For Res. 2017;47(1):134–8. https://doi.org/10.1139/cjfr-2016-0306.
Kaakinen S, Saranpää P, Vapaavuori E. Effects of growth differences due to geographic location and N-fertilisation on wood chemistry of Norway spruce. Trees. 2006;21(2):131. https://doi.org/10.1007/s00468-006-0103-1.
Hart JF. A review of the effects of silviculture on wood quality. The University of British Columbia. Wood . 2010;493:27.
Jozsa L, Brix H. The effects of fertilization and thinning on wood quality of a 24-year-old Douglas-fir stand. Can J For Res. 1989;19(9):1137–45.
Downes GM, Nyakuengama JG, Evans R, Northway R, Blakemore P, Dickson RL, et al. Relationship between wood density, microfibril angle and stiffness in thinned and fertilized Pinus radiata. IAWA J. 2002;23(3):253–65.
Ortega Rodriguez DR, Tomazello-Filho M. Clues to wood quality and production from analyzing ring width and density variabilities of fertilized Pinus taeda trees. New Forest. 2019;50(5):821–43. https://doi.org/10.1007/s11056-018-09702-9.
Mäkinen H, Hynynen J. Wood density and tracheid properties of Scots pine: responses to repeated fertilization and timing of the first commercial thinning. Forestry: Int J For Res. 2014;87(3):437–47.
Mörling T. Evaluation of annual ring width and ring density development following fertilisation and thinning of Scots pine. Ann For Sci. 2002;59(1):29–40.
Kantavichai R, Turnblom EC, Lowell EC. Effects of density control and fertilization on log wood quality from a douglas-fir stand in western oregon, USA. For Sci. 2020;66(2):191–201.
Anttonen S, Manninen A-M, Saranpää P, Kainulainen P, Linder S, Vapaavuori E. Effects of long-term nutrient optimisation on stem wood chemistry in Picea abies. Trees. 2002;16(6):386–94.
Skovsgaard JP, Vanclay JK. Forest site productivity: a review of the evolution of dendrometric concepts for even-aged stands. Forestry: Int J For Res. 2007;81(1):13–31. https://doi.org/10.1093/forestry/cpm041.
Antony F, Jordan L, Schimleck LR, Clark A, Souter RA, Daniels RF. Regional variation in wood modulus of elasticity (stiffness) and modulus of rupture (strength) of planted loblolly pine in the United States. Can J For Res. 2011;41(7):1522–33. https://doi.org/10.1139/x11-064.
Watson P, Bradley M. Canadian pulp fibre morphology: Superiority and considerations for end use potential. For Chron. 2009;85(3):401–8. https://doi.org/10.5558/tfc85401-3.
Watt MS, Trincado G. Development of a generic model describing modulus of elasticity of Pinus radiata in Chile and New Zealand. For Ecol Manag. 2019;453:117583. https://doi.org/10.1016/j.foreco.2019.117583.
Watt MS, Moore JR, Façon J-P, Downes GM, Clinton PW, Coker G, et al. Modelling the influence of stand structural, edaphic and climatic influences on juvenile Pinus radiata dynamic modulus of elasticity. For Ecol Manage. 2006;229(1–3):136–44.
Jordan L, He R, Hall DB, Clark AI, Daniels RF. Variation in loblolly pine ring microfibril angle in the southeastern United States. Wood Fiber Sci. 2007;39(2):352–63.
Palmer DJ, Kimberley MO, Cown DJ, McKinley RB. Assessing prediction accuracy in a regression kriging surface of Pinus radiata outerwood density across New Zealand. For Ecol Manage. 2013;308:9–16.
Rossi S, Cairo E, Krause C, Deslauriers A. Growth and basic wood properties of black spruce along an alti-latitudinal gradient in Quebec. Canada Annals of Forest Science. 2015;72(1):77–87.
Giroud G, Bégin J, Defo M, Ung C-H. Regional variation in wood density and modulus of elasticity of Quebec’s main boreal tree species. For Ecol Manage. 2017;400:289–99.
Nabais C, Hansen JK, David-Schwartz R, Klisz M, López R, Rozenberg P. The effect of climate on wood density: what provenance trials tell us? For Ecol Manage. 2018;408:148–56. https://doi.org/10.1016/j.foreco.2017.10.040.
Pokharel B, Dech JP, Groot A, Pitt D. Ecosite-based predictive modeling of black spruce (Picea mariana) wood quality attributes in boreal Ontario. Can J For Res. 2014;44(5):465–75.
Carson SD, Cown DJ, McKinley RB, Moore JR. Effects of site, silviculture and seedlot on wood density and estimated wood stiffness in radiata pine at mid-rotation. NZ J Forest Sci. 2014;44(1):26. https://doi.org/10.1186/s40490-014-0026-3.
Lenz P, Auty D, Achim A, Beaulieu J, Mackay J. Genetic improvement of white spruce mechanical wood traits—early screening by means of acoustic velocity. Forests. 2013;4(3):575–94.
Hassegawa M, Savard M, Lenz PR, Duchateau E, Gélinas N, Bousquet J, et al. White spruce wood quality for lumber products: priority traits and their enhancement through tree improvement. Forestry: Int J For Res. 2020;93(1):16–37.
Hannrup B, Cahalan C, Chantre G, Grabner M, Karlsson B, Bayon IL et al. Genetic parameters of growth and wood quality traits in Picea abies. Scand J For Res. 2004;19(1):14–29.
Kennedy SG, Cameron AD, Lee S. Genetic relationships between wood quality traits and diameter growth of juvenile core wood in Sitka spruce. Can J For Res. 2013;43(1):1–6.
Li X, Huber DAHA, Powell GLPL, White TLWL, Peter GFPF. Breeding for improved growth and juvenile corewood stiffness in slash pine. Can J For Res. 2007;37(10):1886–93. https://doi.org/10.1139/x07-043.
Weng YH, Lu P, Adams GW, Fullarton MS, Tosh KJ. Genetic parameters of growth and stem quality traits for jack pine second-generation progeny tested in New Brunswick. Can J For Res. 2015;45(1):36–43. https://doi.org/10.1139/cjfr-2014-0106.
Lenz P, Cloutier A, MacKay J, Beaulieu J. Genetic control of wood properties in Picea glauca—an analysis of trends with cambial age. Can J For Res. 2010;40(4):703–15.
Corriveau A, Beaulieu J, Daoust G. Heritability and genetic correlations of wood characters of Upper Ottawa Valley white spruce populations grown in Quebec. For Chron. 1991;67(6):698–705.
Chen Z-Q, Karlsson B, Mörling T, Olsson L, Mellerowicz EJ, Wu HX, et al. Genetic analysis of fiber dimensions and their correlation with stem diameter and solid-wood properties in Norway spruce. Tree Genet Genomes. 2016;12(6):1–12.
Desponts M, Perron M, DeBlois J. Rapid assessment of wood traits for large-scale breeding selection in Picea mariana [Mill.] BSP. Annals For Sci. 2017;74(3):53.
Jayawickrama K. Genetic parameter estimates for radiata pine in New Zealand and New South Wales: a synthesis of results. Silvae Genetica. 2001;50(2):45–53.
Beaulieu J, Doerksen T, Clément S, MacKay J, Bousquet J. Accuracy of genomic selection models in a large population of open-pollinated families in white spruce. Heredity. 2014;113(4):343–52.
Global_Climate_Report. 2021. https://www.ncdc.noaa.gov/sotc/global/202104. Accessed 2021.
Menzel A, Fabian P. Growing season extended in Europe. Nature. 1999;397(6721):659. https://doi.org/10.1038/17709.
Morin X, Lechowicz MJ, Augspurger C, O’keefe J, Viner D, Chuine I. Leaf phenology in 22 North American tree species during the 21st century. Glob Change Biol. 2009;15(4):961–75.
Deslauriers A, Morin H, Begin Y. Cellular phenology of annual ring formation of Abies balsamea in the Quebec boreal forest (Canada). Can J For Res. 2003;33(2):190–200.
Auty D, Achim A. The relationship between standing tree acoustic assessment and timber quality in Scots pine and the practical implications for assessing timber quality from naturally regenerated stands. Forestry. 2008;81(4):475–87.
Piao S, Liu Q, Chen A, Janssens IA, Fu Y, Dai J, et al. Plant phenology and global climate change: Current progresses and challenges. Glob Change Biol. 2019;25(6):1922–40.
Babst F, Bouriaud O, Poulter B, Trouet V, Girardin MP, Frank DC. Twentieth century redistribution in climatic drivers of global tree growth. Sci Adv. 2019;5(1):eaat4313. The paper highlights that water availability has replaced energy as the dominant limiting factor of tree growth across large portions of the boreal zone. This evidence suggests that drought is becoming the dominant limitation of tree growth worldwide.
Kurz-Besson CB, Lousada JL, Gaspar MJ, Correia IE, David TS, Soares PMM et al. Effects of recent minimum temperature and water deficit increases on Pinus pinaster radial growth and wood density in southern Portugal. Front Plant Sci. 2016;7(1170). https://doi.org/10.3389/fpls.2016.01170
Von Arx G, Archer SR, Hughes MK. Long-term functional plasticity in plant hydraulic architecture in response to supplemental moisture. Ann Bot. 2012;109(6):1091–100.
Primicia I, Camarero JJ, Imbert JB, Castillo FJ. Effects of thinning and canopy type on growth dynamics of Pinus sylvestris: inter-annual variations and intra-annual interactions with microclimate. Eur J Forest Res. 2013;132(1):121–35.
Björklund J, Seftigen K, Schweingruber F, Fonti P, von Arx G, Bryukhanova MV, et al. Cell size and wall dimensions drive distinct variability of earlywood and latewood density in Northern Hemisphere conifers. New Phytol. 2017;216(3):728–40.
McDowell N, Pockman WT, Allen CD, Breshears DD, Cobb N, Kolb T, et al. Mechanisms of plant survival and mortality during drought: why do some plants survive while others succumb to drought? New Phytol. 2008;178(4):719–39.
Pittermann J, Sperry JS, Hacke UG, Wheeler JK, Sikkema EH. Inter-tracheid pitting and the hydraulic efficiency of conifer wood: the role of tracheid allometry and cavitation protection. Am J Bot. 2006;93(9):1265–73.
Fleming RA. Climate change and insect disturbance regimes in Canada’s boreal forests. World Resour Rev. 2000;12(3):521–48.
Seidl R, Thom D, Kautz M, Martin-Benito D, Peltoniemi M, Vacchiano G, et al. Forest disturbances under climate change. Nat Clim Chang. 2017;7(6):395–402.
Jactel H, Koricheva J, Castagneyrol B. Responses of forest insect pests to climate change: not so simple. Curr Opin Insect Sci. 2019;35:103–8. https://doi.org/10.1016/j.cois.2019.07.010.
Achim A, Moreau G, Coops NC, Axelson JN, Barrette J, Bédard S et al. The changing culture of silviculture. Forestry. 2022;95(2):143–52.
Gardiner B. Wind damage to forests and trees: a review with an emphasis on planted and managed forests. J For Res. 2021;26(4):248–66.
Bauce É, Crépin M, Carisey N. Spruce budworm growth, development and food utilization on young and old balsam fir trees. Oecologia. 1994;97(4):499–507.
Bennett AC, McDowell NG, Allen CD, Anderson-Teixeira KJ. Larger trees suffer most during drought in forests worldwide. Nature Plants. 2015;1(10):15139. https://doi.org/10.1038/nplants.2015.139.
Kim D-W, Chung W, Lee B. Exploring tree crown spacing and slope interaction effects on fire behavior with a physics-based fire model. For Sci Technol. 2016;12(4):167–75. https://doi.org/10.1080/21580103.2016.1144541.
Büntgen U, Krusic PJ, Piermattei A, Coomes DA, Esper J, Myglan VS, et al. Limited capacity of tree growth to mitigate the global greenhouse effect under predicted warming. Nat Commun. 2019;10(1):1–6.
Waring RH, Pitman GB. Modifying lodgepole pine stands to change susceptibility to mountain pine beetle attack. Ecology. 1985;66(3):889–97. https://doi.org/10.2307/1940551.
Dodds KJ, Cooke RR, Hanavan RP. The Effects of Silvicultural Treatment on Sirex noctilio Attacks and Tree Health in Northeastern United States. Forests. 2014;5(11):2810–24.
Bauce É, Fuentealba A. Interactions between stand thinning, site quality and host tree species on spruce budworm biological performance and host tree resistance over a 6 year period after thinning. For Ecol Manage. 2013;304:212–23. https://doi.org/10.1016/j.foreco.2013.05.008.
Moreau G, Chagnon C, Achim A, Caspersen J, D’Orangeville L, Sánchez-Pinillos M et al. Opportunities and limitations of thinning to increase resistance and resilience of trees and forests to global change. Forestry. 2022;95(5):595–615. The paper reviews recent literature to identify the positive and negative effects of thinning on both stand- and tree-level resistance and resilience to four stressors that are expected to increase in frequency and/or severity due to global change: drought, fire, insects and pathogens, and wind.
Peltola H, Gardiner B, Nicoll B. Mechanics of wind damage. Agric For Meteorol. 2013;151:328–44.
Gardiner B, Barnett J, Saranpää P, Gril J. The biology of reaction wood. Springer; 2014.
Lindroth A, Lagergren F, Grelle A, Klemedtsson L, Langvall O, Weslien P, et al. Storms can cause Europe-wide reduction in forest carbon sink. Glob Change Biol. 2009;15(2):346–55.
Desch HE, Dinwoodie JM. Timber: structure, properties, conversion and use. Macmillan International Higher Education; 1996.
Lenz PRN, Nadeau S, Mottet M-J, Perron M, Isabel N, Beaulieu J, et al. Multi-trait genomic selection for weevil resistance, growth, and wood quality in Norway spruce. Evol Appl. 2020;13(1):76–94. https://doi.org/10.1111/eva.12823.
Sniezko RA, Koch J. Breeding trees resistant to insects and diseases: putting theory into application. Biol Invasions. 2017;19(11):3377–400. https://doi.org/10.1007/s10530-017-1482-5.
Naidoo S, Slippers B, Plett JM, Coles D, Oates CN. The road to resistance in forest trees. Front Plant Sci. 2019;10:273.
Drew DM, Downes GM, Seifert T, Eckes-Shepard A, Achim A. A Review of Progress and Applications in Wood Quality Modelling. Current Forestry Reports; 2022. p. 1–16. The paper reviews models that predict wood quality in standing trees. They propose a simple classification of wood quality models into two broad groupings: fully empirical and process-based. Comprehensive summaries of a wide range of published models in both categories are given.
Lachenbruch B, Moore JR, Evans R. Radial variation in wood structure and function in woody plants, and hypotheses for its occurrence. Size-and age-related changes in tree structure and function. Springer; 2011. pp. 121–64.
Auty D, Achim A, Macdonald E, Cameron AD, Gardiner BA. Models for predicting wood density variation in Scots pine. Forestry: Int J For Res. 2014;87(3):449–58.
Auty D, Achim A, Macdonald E, Cameron AD, Gardiner BA. Models for predicting clearwood mechanical properties of Scots pine. For Sci. 2016;62(4):403–13.
Gardiner B, Leban J-M, Auty D, Simpson H. Models for predicting wood density of British-grown Sitka spruce. Forestry. 2011;84(2):119–32.
Leban J-M, Haines DW. The modulus of elasticity of hybrid larch predicted by density, rings per centimeter, and age. Wood Fiber Sci. 1999;31(4):394–402.
Schimleck LR, Antony F, Mora C, Dahlen J. Mapping and modeling within-tree variation for loblolly pine pulp yield and lignin content. SN Appl Sci. 2021;3(4):468. https://doi.org/10.1007/s42452-021-04443-5.
Houllier F, Leban J-M, Colin F. Linking growth modelling to timber quality assessment for Norway spruce. For Ecol Manage. 1995;74(1–3):91–102.
Bontemps J-D, Esper J. Statistical modelling and RCS detrending methods provide similar estimates of long-term trend in radial growth of common beech in north-eastern France. Dendrochronologia. 2011;29(2):99–107.
Auty D, Moore J, Achim A, Lyon A, Mochan S, Gardiner B. Effects of early respacing on the density and microfibril angle of Sitka spruce wood. Forestry: Int J For Res. 2018;91(3):307–19.
Soro A, Lenz P, Hassegawa M, Roussel J-R, Bousquet J, Achim A. Genetic influence on components of wood density variation in white spruce. Forestry: Int J For Res. 2021.
Drew DM, Allen K, Downes GM, Evans R, Battaglia M, Baker P. Wood properties in a long-lived conifer reveal strong climate signals where ring-width series do not. Tree Physiol. 2013;33(1):37–47.
Vaughan D, Auty D, Kolb T, Dahlen J, Sánchez Meador AJ, Mackes KH. Wood density variation in naturally regenerated stands of Pinus ponderosa in northern Arizona, USA. Can J For Res. 2021;51(4):583–94.
Franceschini T, Longuetaud F, Bontemps J-D, Bouriaud O, Caritey B-D, Leban J-M. Effect of ring width, cambial age, and climatic variables on the within-ring wood density profile of Norway spruce Picea abies (L.) Karst. Trees. 2013;27(4):913–25.
Wood LJ, Smith DJ, Hartley ID. Predicting softwood quality attributes from climate data in interior British Columbia, Canada. For Ecol Manag. 2016;361:81–9.
Xiang W, Auty D, Franceschini T, Leitch M, Achim A. Wood density-climate relationships are mediated by dominance class in black spruce (Picea mariana (Mill.) BSP). Forests. 2014;5(6):1163–84.
Candel-Pérez D, Lo Y-H, Blanco JA, Chiu C-M, Camarero JJ, González de Andrés E, et al. Drought-induced changes in wood density are not prevented by thinning in scots pine stands. Forests. 2018;9(1):4.
Benomar L, Bousquet J, Perron M, Beaulieu J, Lamara M. Tree maladaptation under mid-latitude early spring warming and late cold spell: implications for assisted migration. Front Plant Sci. 2022:2086.
Payette S, Fortin M-J, Morneau C. The recent sugar maple decline in southern Quebec: probable causes deduced from tree rings. Can J For Res. 1996;26(6):1069–78.
Moreau G, Chagnon C, Auty D, Caspersen J, Achim A. Impacts of climatic variation on the growth of black spruce across the forest-tundra ecotone: positive effects of warm growing seasons and heat waves are offset by late spring frosts. Front For Glob Change. 2020;3:613523.
Babst F, Bodesheim P, Charney N, Friend AD, Girardin MP, Klesse S, et al. When tree rings go global: Challenges and opportunities for retro-and prospective insight. Quatern Sci Rev. 2018;197:1–20.
West G, Moore J, Shula R, Harrington J, Snook J, Gordon J et al. Forest management DSS development in New Zealand. Implementation of DSS tools into the forestry practice. 2013:153–63.
Anderegg WRL, Hicke JA, Fisher RA, Allen CD, Aukema J, Bentz B, et al. Tree mortality from drought, insects, and their interactions in a changing climate. New Phytol. 2015;208(3):674–83. https://doi.org/10.1111/nph.13477.
Dufour-Kowalski S, Courbaud B, Dreyfus P, Meredieu C, De Coligny F. Capsis: an open software framework and community for forest growth modelling. Ann For Sci. 2012;69(2):221–33.
Jacquin P, Mothe F, Longuetaud F, Billard A, Kerfriden B, Leban J-M. CarDen: a software for fast measurement of wood density on increment cores by CT scanning. Comput Electron Agric. 2019;156:606–17.
Freyburger C, Longuetaud F, Mothe F, Constant T, Leban J-M. Measuring wood density by means of X-ray computer tomography. Ann For Sci. 2009;66(8):804.
Dierick M, Van Loo D, Masschaele B, Van den Bulcke J, Van Acker J, Cnudde V, et al. Recent micro-CT scanner developments at UGCT. Nucl Instrum Methods Phys Res, Sect B. 2014;324:35–40.
Kharrat W, Koubaa A, Khlif M, Bradai C. Intra-ring wood density and dynamic modulus of elasticity profiles for black spruce and jack pine from X-ray densitometry and ultrasonic wave velocity measurement. Forests. 2019;10(7):569.
Luther JE, Skinner R, Fournier RA, van Lier OR, Bowers WW, Coté J-F, et al. Predicting wood quantity and quality attributes of balsam fir and black spruce using airborne laser scanner data. Forestry. 2014;87(2):313–26.
Hilker T, Frazer GW, Coops NC, Wulder MA, Newnham GJ, Stewart JD, et al. Prediction of wood fiber attributes from LiDAR-derived forest canopy indicators. Forest Sci. 2013;59(2):231–42.
Van Leeuwen M, Hilker T, Coops NC, Frazer G, Wulder MA, Newnham GJ, et al. Assessment of standing wood and fiber quality using ground and airborne laser scanning: a review. For Ecol Manage. 2011;261(9):1467–78.
Pyörälä J, Saarinen N, Kankare V, Coops NC, Liang X, Wang Y, et al. Variability of wood properties using airborne and terrestrial laser scanning. Remote Sens Environ. 2019;235:111474.
Barton CV. Advances in remote sensing of plant stress. Plant Soil. 2012;354(1):41–4.
Hilker T, Coops NC, Hall FG, Nichol CJ, Lyapustin A, Black TA et al. Inferring terrestrial photosynthetic light use efficiency of temperate ecosystems from space. J Geophys Res: Biogeosci. 2011;116(G3).
Dickinson Y, Wang X, Wiedenbeck J, Ross RJ. Effects of silviculture practices on engineering properties of northern hardwood species of the Great Lakes Region: a literature review. Gen Tech Rep FPL-GTR-269 Madison, WI: US Department of Agriculture, Forest Service, Forest Products Laboratory, 2019;17:269.
Moore JR, Cown DJ. Corewood (Juvenile Wood) and Its Impact on Wood Utilisation. Current Forestry Reports. 2017;3(2):107–18. https://doi.org/10.1007/s40725-017-0055-2. This review focuses on describing the characteristics of corewood, the factors affecting its occurrence within a tree and the implications for wood utilisation.
Briggs D. Enhancing forest value productivity through fiber quality. J Forest. 2010;108(4):174–82.
Cave I, Walker J. Stiffness of wood in fast-grown plantation softwoods: the influence of microfibril angle. For Prod J. 1994;44(5):43.
Giroud G, Defo M, Bégin J. Determination of radial profiles of wood properties using a near infrared scanning system. J Near Infrared Spectrosc. 2021;29(1):24–32.
Auty D, Gardiner BA, Achim A, Moore JR, Cameron AD. Models for predicting microfibril angle variation in Scots pine. Ann For Sci. 2013;70(2):209–18.
Moore JR, Cown DJ, McKinley RB. Modelling microfibril angle variation in New Zealand-grown radiata pine. NZ J Forest Sci. 2014;44(1):1–11.
Telewski F. Structure and function of flexure wood in Abies fraseri. Tree Physiol. 1989;5(1):113–21.
Wang S-Y, Chen J-H, Tsai M-J, Lin C-J, Yang T-H. Grading of softwood lumber using non-destructive techniques. J Mater Process Technol. 2008;208(1):149–58. https://doi.org/10.1016/j.jmatprotec.2007.12.105.
Moore JR, Lyon AJ, Searles GJ, Lehneke SA, Ridley-Ellis DJ. Within-and between-stand variation in selected properties of Sitka spruce sawn timber in the UK: implications for segregation and grade recovery. Ann For Sci. 2013;70(4):403–15.
Ridley-Ellis D, Gil-Moreno D, Harte AM. Strength grading of timber in the UK and Ireland in. Int Wood Prod J. 2021;2022:1–10.
Acknowledgements
This work was conducted as part of project number 142332173 led by Julie Barrette at the Direction de la Recherche Forestière (ministère des Ressources Naturelles et des Forêts, Québec, Canada) and took part of the network Matériaux Renouvelables Québec. We would like to thank Jean Noël for the production of Fig. 1 as well as two anonymous reviewers for providing insightful suggestions on a previous version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Wood Structure and Function
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Barrette, J., Achim, A. & Auty, D. Impact of Intensive Forest Management Practices on Wood Quality from Conifers: Literature Review and Reflection on Future Challenges. Curr Forestry Rep 9, 101–130 (2023). https://doi.org/10.1007/s40725-023-00181-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40725-023-00181-6