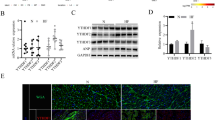
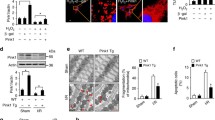
Abstract
Regulation of RNA stability and translation by RNA-binding proteins (RBPs) is a crucial process altering gene expression. Musashi family of RBPs comprising Msi1 and Msi2 is known to control RNA stability and translation. However, despite the presence of MSI2 in the heart, its function remains largely unknown. Here, we aim to explore the cardiac functions of MSI2. We confirmed the presence of MSI2 in the adult mouse, rat heart, and neonatal rat cardiomyocytes. Furthermore, Msi2 was significantly enriched in the heart cardiomyocyte fraction. Next, using RNA-seq data and isoform-specific PCR primers, we identified Msi2 isoforms 1, 4, and 5, and two novel putative isoforms labeled as Msi2 6 and 7 to be expressed in the heart. Overexpression of Msi2 isoforms led to cardiac hypertrophy in cultured cardiomyocytes. Additionally, Msi2 exhibited a significant increase in a pressure-overload model of cardiac hypertrophy. We selected isoforms 4 and 7 to validate the hypertrophic effects due to their unique alternative splicing patterns. AAV9-mediated overexpression of Msi2 isoforms 4 and 7 in murine hearts led to cardiac hypertrophy, dilation, heart failure, and eventually early death, confirming a pathological function for Msi2. Using global proteomics, gene ontology, transmission electron microscopy, seahorse, and transmembrane potential measurement assays, increased MSI2 was found to cause mitochondrial dysfunction in the heart. Mechanistically, we identified Cluh and Smyd1 as direct downstream targets of Msi2. Overexpression of Cluh and Smyd1 inhibited Msi2-induced cardiac malfunction and mitochondrial dysfunction. Collectively, we show that Msi2 induces hypertrophy, mitochondrial dysfunction, and heart failure.









Similar content being viewed by others
Data availability
All the necessary data has been provided within the manuscript. Any further data are available from the corresponding author upon request.
Abbreviations
- RBP:
-
RNA-binding protein
- AAV:
-
Adeno-associated virus
- GFP:
-
Green fluorescent protein
- UTR:
-
Untranslated region
- DNA:
-
Deoxyribonucleic acid
- RNA:
-
Ribonucleic acid
- rRNA:
-
Ribosomal RNA
- tRNA:
-
Transfer RNA
- mRNA:
-
Messenger RNA
- NCBI:
-
National center for biotechnology information
- PCR:
-
Polymerase chain reaction
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- TEM:
-
Transmission electron microscope
- RRM:
-
RNA recognition motifs
- LC–MS/MS:
-
Liquid chromatography with tandem mass spectrometry
- GO:
-
Gene ontology
- TCA:
-
Tricarboxylic acid
- ARE:
-
Adenylate-uridylate-rich elements
- TMRE:
-
Tetramethylrhodamine, ethyl ester
- TAC:
-
Trans-aortic constriction
- FPKM:
-
Fragment per kilobase of transcript per million read pairs
- MOI:
-
Multiplicity of Infection
- ChIP:
-
Chromatin immunoprecipitation
- PFA:
-
Paraformaldehyde
- CVD:
-
Cardiovascular diseases
- HF:
-
Heart failure
- FBS:
-
Fetal bovine serum
References
Ackers-Johnson M, Li PY, Holmes AP, O’Brien SM, Pavlovic D, Foo RS (2016) A simplified, langendorff-free method for concomitant isolation of viable cardiac myocytes and nonmyocytes from the adult mouse heart. Circ Res 119:909–920. https://doi.org/10.1161/CIRCRESAHA.116.309202
Bennett CG, Riemondy K, Chapnick DA, Bunker E, Liu X, Kuersten S, Yi R (2016) Genome-wide analysis of Musashi-2 targets reveals novel functions in governing epithelial cell migration. Nucleic Acids Res 44:3788–3800. https://doi.org/10.1093/nar/gkw207
Bhattacharjee A, Hasanain M, Kathuria M, Singh A, Datta D, Sarkar J, Mitra K (2018) Ormeloxifene-induced unfolded protein response contributes to autophagy-associated apoptosis via disruption of Akt/mTOR and activation of JNK. Sci Rep 8:2303. https://doi.org/10.1038/s41598-018-20541-8
Chang KT, Cheng CF, King PC, Liu SY, Wang GS (2017) CELF1 Mediates connexin 43 mRNA degradation in dilated cardiomyopathy. Circ Res 121:1140–1152. https://doi.org/10.1161/CIRCRESAHA.117.311281
Chen S, Zhou Y, Chen Y, Gu J (2018) fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34:i884–i890. https://doi.org/10.1093/bioinformatics/bty560
Choudhury NR, de Lima AF, de Andres-Aguayo L, Graf T, Caceres JF, Rappsilber J, Michlewski G (2013) Tissue-specific control of brain-enriched miR-7 biogenesis. Genes Dev 27:24–38. https://doi.org/10.1101/gad.199190.112
Corbett AH (2018) Post-transcriptional regulation of gene expression and human disease. Curr Opin Cell Biol 52:96–104. https://doi.org/10.1016/j.ceb.2018.02.011
Frangogiannis NG (2021) Cardiac fibrosis. Cardiovasc Res 117:1450–1488. https://doi.org/10.1093/cvr/cvaa324
Franklin S, Kimball T, Rasmussen TL, Rosa-Garrido M, Chen H, Tran T, Miller MR, Gray R, Jiang S, Ren S, Wang Y, Tucker HO, Vondriska TM (2016) The chromatin-binding protein Smyd1 restricts adult mammalian heart growth. Am J Physiol Heart Circ Physiol 311:H1234–H1247. https://doi.org/10.1152/ajpheart.00235.2016
Froese N, Cordero J, Abouissa A, Trogisch FA, Grein S, Szaroszyk M, Wang Y, Gigina A, Korf-Klingebiel M, Bosnjak B, Davenport CF, Wiehlmann L, Geffers R, Riechert E, Jurgensen L, Boileau E, Lin Y, Dieterich C, Forster R, Bauersachs J, Ola R, Dobreva G, Volkers M, Heineke J (2022) Analysis of myocardial cellular gene expression during pressure overload reveals matrix based functional intercellular communication. IScience 25:103965. https://doi.org/10.1016/j.isci.2022.103965
Gao C, Wang Y (2020) mRNA metabolism in cardiac development and disease: life after transcription. Physiol Rev 100:673–694. https://doi.org/10.1152/physrev.00007.2019
Gao J, Schatton D, Martinelli P, Hansen H, Pla-Martin D, Barth E, Becker C, Altmueller J, Frommolt P, Sardiello M, Rugarli EI (2014) CLUH regulates mitochondrial biogenesis by binding mRNAs of nuclear-encoded mitochondrial proteins. J Cell Biol 207:213–223. https://doi.org/10.1083/jcb.201403129
Guo QQ, Gao J, Wang XW, Yin XL, Zhang SC, Li X, Chi LL, Zhou XM, Wang Z, Zhang QY (2020) RNA-binding protein MSI2 binds to miR-301a-3p and facilitates its distribution in mitochondria of endothelial cells. Front Mol Biosci 7:609828. https://doi.org/10.3389/fmolb.2020.609828
Gupta SK, Garg A, Avramopoulos P, Engelhardt S, Streckfuss-Bomeke K, Batkai S, Thum T (2019) miR-212/132 cluster modulation prevents doxorubicin-mediated atrophy and cardiotoxicity. Mol Ther 27:17–28. https://doi.org/10.1016/j.ymthe.2018.11.004
Hentze MW, Castello A, Schwarzl T, Preiss T (2018) A brave new world of RNA-binding proteins. Nat Rev Mol Cell Biol 19:327–341. https://doi.org/10.1038/nrm.2017.130
Katz Y, Li F, Lambert NJ, Sokol ES, Tam WL, Cheng AW, Airoldi EM, Lengner CJ, Gupta PB, Yu Z, Jaenisch R, Burge CB (2014) Musashi proteins are post-transcriptional regulators of the epithelial-luminal cell state. Elife 3:e03915. https://doi.org/10.7554/eLife.03915
Kharas MG, Lengner CJ (2017) Stem cells, cancer, and musashi in blood and guts. Trends Cancer 3:347–356. https://doi.org/10.1016/j.trecan.2017.03.007
Kim D, Paggi JM, Park C, Bennett C, Salzberg SL (2019) Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat Biotechnol 37:907–915. https://doi.org/10.1038/s41587-019-0201-4
Kimura T, Ferran B, Tsukahara Y, Shang Q, Desai S, Fedoce A, Pimentel DR, Luptak I, Adachi T, Ido Y, Matsui R, Bachschmid MM (2019) Production of adeno-associated virus vectors for in vitro and in vivo applications. Sci Rep 9:13601. https://doi.org/10.1038/s41598-019-49624-w
Li M, Li AQ, Zhou SL, Lv H, Wei P, Yang WT (2020) RNA-binding protein MSI2 isoforms expression and regulation in progression of triple-negative breast cancer. J Exp Clin Cancer Res 39:92. https://doi.org/10.1186/s13046-020-01587-x
Li X, La Salvia S, Liang Y, Adamiak M, Kohlbrenner E, Jeong D, Chepurko E, Ceholski D, Lopez-Gordo E, Yoon S, Mathiyalagan P, Agarwal N, Jha D, Lodha S, Daaboul G, Phan A, Raisinghani N, Zhang S, Zangi L, Gonzalez-Kozlova E, Dubois N, Dogra N, Hajjar RJ, Sahoo S (2023) Extracellular vesicle-encapsulated adeno-associated viruses for therapeutic gene delivery to the heart. Circulation 148:405–425. https://doi.org/10.1161/CIRCULATIONAHA.122.063759
Nakamura M, Okano H, Blendy JA, Montell C (1994) Musashi, a neural RNA-binding protein required for Drosophila adult external sensory organ development. Neuron 13:67–81. https://doi.org/10.1016/0896-6273(94)90460-x
Oka SI, Sabry AD, Horiuchi AK, Cawley KM, O’Very SA, Zaitsev MA, Shankar TS, Byun J, Mukai R, Xu X, Torres NS, Kumar A, Yazawa M, Ling J, Taleb I, Saijoh Y, Drakos SG, Sadoshima J, Warren JS (2020) Perm1 regulates cardiac energetics as a downstream target of the histone methyltransferase Smyd1. PLoS ONE 15:e0234913. https://doi.org/10.1371/journal.pone.0234913
Paulus MG, Renner K, Nickel AG, Brochhausen C, Limm K, Zugner E, Baier MJ, Pabel S, Wallner S, Birner C, Luchner A, Magnes C, Oefner PJ, Stark KJ, Wagner S, Maack C, Maier LS, Streckfuss-Bomeke K, Sossalla S, Dietl A (2022) Tachycardiomyopathy entails a dysfunctional pattern of interrelated mitochondrial functions. Basic Res Cardiol 117:45. https://doi.org/10.1007/s00395-022-00949-0
Peoples JN, Saraf A, Ghazal N, Pham TT, Kwong JQ (2019) Mitochondrial dysfunction and oxidative stress in heart disease. Exp Mol Med 51:1–13. https://doi.org/10.1038/s12276-019-0355-7
Pertea M, Pertea GM, Antonescu CM, Chang TC, Mendell JT, Salzberg SL (2015) StringTie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol 33:290–295. https://doi.org/10.1038/nbt.3122
Pun-Garcia A, Clemente-Moragon A, Villena-Gutierrez R, Gomez M, Sanz-Rosa D, Diaz-Guerra A, Prados B, Medina JP, Monto F, Ivorra MD, Marquez-Lopez C, Cannavo A, Bernal JA, Koch WJ, Fuster V, de la Pompa JL, Oliver E, Ibanez B (2022) Beta-3 adrenergic receptor overexpression reverses aortic stenosis-induced heart failure and restores balanced mitochondrial dynamics. Basic Res Cardiol 117:62. https://doi.org/10.1007/s00395-022-00966-z
Riechert E, Kmietczyk V, Stein F, Schwarzl T, Sekaran T, Jurgensen L, Kamuf-Schenk V, Varma E, Hofmann C, Rettel M, Gur K, Olschlager J, Kuhl F, Martin J, Ramirez-Pedraza M, Fernandez M, Doroudgar S, Mendez R, Katus HA, Hentze MW, Volkers M (2021) Identification of dynamic RNA-binding proteins uncovers a Cpeb4-controlled regulatory cascade during pathological cell growth of cardiomyocytes. Cell Rep 35:109100. https://doi.org/10.1016/j.celrep.2021.109100
Roth GA, Mensah GA, Johnson CO, Addolorato G, Ammirati E, Baddour LM, Barengo NC, Beaton AZ, Benjamin EJ, Benziger CP, Bonny A, Brauer M, Brodmann M, Cahill TJ, Carapetis J, Catapano AL, Chugh SS, Cooper LT, Coresh J, Criqui M, DeCleene N, Eagle KA, Emmons-Bell S, Feigin VL, Fernandez-Sola J, Fowkes G, Gakidou E, Grundy SM, He FJ, Howard G, Hu F, Inker L, Karthikeyan G, Kassebaum N, Koroshetz W, Lavie C, Lloyd-Jones D, Lu HS, Mirijello A, Temesgen AM, Mokdad A, Moran AE, Muntner P, Narula J, Neal B, Ntsekhe M, Moraes de Oliveira G, Otto C, Owolabi M, Pratt M, Rajagopalan S, Reitsma M, Ribeiro ALP, Rigotti N, Rodgers A, Sable C, Shakil S, Sliwa-Hahnle K, Stark B, Sundstrom J, Timpel P, Tleyjeh IM, Valgimigli M, Vos T, Whelton PK, Yacoub M, Zuhlke L, Murray C, Fuster V, Group G-N-JGBoCDW (2020) Global burden of cardiovascular diseases and risk factors, 1990–2019: update from the GBD study. J Am Coll Cardiol. 76(2982):3021. https://doi.org/10.1016/j.jacc.2020.11.010
Sabater-Arcis M, Bargiela A, Moreno N, Poyatos-Garcia J, Vilchez JJ, Artero R (2021) Musashi-2 contributes to myotonic dystrophy muscle dysfunction by promoting excessive autophagy through miR-7 biogenesis repression. Mol Ther Nucleic Acids 25:652–667. https://doi.org/10.1016/j.omtn.2021.08.010
Sabbah HN (2020) Targeting the mitochondria in heart failure: a translational perspective. JACC Basic Transl Sci 5:88–106. https://doi.org/10.1016/j.jacbts.2019.07.009
Sakakibara S, Imai T, Hamaguchi K, Okabe M, Aruga J, Nakajima K, Yasutomi D, Nagata T, Kurihara Y, Uesugi S, Miyata T, Ogawa M, Mikoshiba K, Okano H (1996) Mouse-Musashi-1, a neural RNA-binding protein highly enriched in the mammalian CNS stem cell. Dev Biol 176:230–242. https://doi.org/10.1006/dbio.1996.0130
Sakakibara S, Nakamura Y, Satoh H, Okano H (2001) Rna-binding protein Musashi2: developmentally regulated expression in neural precursor cells and subpopulations of neurons in mammalian CNS. J Neurosci 21:8091–8107
Scarpulla RC (2012) Nucleus-encoded regulators of mitochondrial function: integration of respiratory chain expression, nutrient sensing and metabolic stress. Biochim Biophys Acta 1819:1088–1097. https://doi.org/10.1016/j.bbagrm.2011.10.011
Schatton D, Pla-Martin D, Marx MC, Hansen H, Mourier A, Nemazanyy I, Pessia A, Zentis P, Corona T, Kondylis V, Barth E, Schauss AC, Velagapudi V, Rugarli EI (2017) CLUH regulates mitochondrial metabolism by controlling translation and decay of target mRNAs. J Cell Biol 216:675–693. https://doi.org/10.1083/jcb.201607019
Schimmel K, Jung M, Foinquinos A, Jose GS, Beaumont J, Bock K, Grote-Levi L, Xiao K, Bar C, Pfanne A, Just A, Zimmer K, Ngoy S, Lopez B, Ravassa S, Samolovac S, Janssen-Peters H, Remke J, Scherf K, Dangwal S, Piccoli MT, Kleemiss F, Kreutzer FP, Kenneweg F, Leonardy J, Hobuss L, Santer L, Do QT, Geffers R, Braesen JH, Schmitz J, Brandenberger C, Muller DN, Wilck N, Kaever V, Bahre H, Batkai S, Fiedler J, Alexander KM, Wertheim BM, Fisch S, Liao R, Diez J, Gonzalez A, Thum T (2020) Natural compound library screening identifies new molecules for the treatment of cardiac fibrosis and diastolic dysfunction. Circulation 141:751–767. https://doi.org/10.1161/CIRCULATIONAHA.119.042559
Schoenberg DR, Maquat LE (2012) Regulation of cytoplasmic mRNA decay. Nat Rev Genet 13:246–259. https://doi.org/10.1038/nrg3160
Shi R, Ying S, Li Y, Zhu L, Wang X, Jin H (2021) Linking the YTH domain to cancer: the importance of YTH family proteins in epigenetics. Cell Death Dis 12:346. https://doi.org/10.1038/s41419-021-03625-8
Suresh Babu S, Joladarashi D, Jeyabal P, Thandavarayan RA, Krishnamurthy P (2015) RNA-stabilizing proteins as molecular targets in cardiovascular pathologies. Trends Cardiovasc Med 25:676–683. https://doi.org/10.1016/j.tcm.2015.02.006
Warren JS, Tracy CM, Miller MR, Makaju A, Szulik MW, Oka SI, Yuzyuk TN, Cox JE, Kumar A, Lozier BK, Wang L, Llana JG, Sabry AD, Cawley KM, Barton DW, Han YH, Boudina S, Fiehn O, Tucker HO, Zaitsev AV, Franklin S (2018) Histone methyltransferase Smyd1 regulates mitochondrial energetics in the heart. Proc Natl Acad Sci USA 115:E7871–E7880. https://doi.org/10.1073/pnas.1800680115
Yang D, Liu HQ, Liu FY, Guo Z, An P, Wang MY, Yang Z, Fan D, Tang QZ (2021) Mitochondria in pathological cardiac hypertrophy research and therapy. Front Cardiovasc Med 8:822969. https://doi.org/10.3389/fcvm.2021.822969
Yang W, Yang L, Wang J, Zhang Y, Li S, Yin Q, Suo J, Ma R, Ye Y, Cheng H, Li J, Hui J, Hu P (2022) Msi2-mediated MiR7a-1 processing repression promotes myogenesis. J Cachexia Sarcopenia Muscle 13:728–742. https://doi.org/10.1002/jcsm.12882
Zhang Y, Si Y, Ma N, Mei J (2015) The RNA-binding protein PCBP2 inhibits Ang II-induced hypertrophy of cardiomyocytes though promoting GPR56 mRNA degeneration. Biochem Biophys Res Commun 464:679–684. https://doi.org/10.1016/j.bbrc.2015.06.139
Zhou A, Shi G, Kang GJ, Xie A, Liu H, Jiang N, Liu M, Jeong EM, Dudley SC Jr (2018) RNA binding protein, hur, regulates SCN5A expression through stabilizing MEF2C transcription factor mRNA. J Am Heart Assoc. https://doi.org/10.1161/JAHA.117.007802
Acknowledgements
We want to thank Dr. Rajdeep Guha, Head of Animal Facility, CSIR-CDRI, for his help with animal experiments. We want to thank Director CSIR-CDRI, Dr. Manoj Kumar Barthwal, Dr. Amit Lahiri, Dr. Chandra Prakash Pandey, Dr. Hobby Aggarwal, Dr. Jayanta Sarkar, and Mr. Ajay Singh from CSIR-CDRI, for their help with the instruments and training. We acknowledge the FACS facility, CSIR-CDRI. We acknowledge the valuable suggestions of Dr. Regalla Kumarswamy, CSIR- Centre for Cellular and Molecular Biology, Hyderabad, India, Dr. Marisol Ruiz- Meana, Valld’Hebron Hospital Universitari, Barcelona, Spain, and Dr. Gaurav Ahuja, Indraprastha Institute of Information Technology, Delhi, India. This manuscript has CSIR-CDRI communication number 10680.
Funding
This work was funded by Ramalingaswami Re-entry Fellowship (BT/RLF/re-entry/14/2019) from the Department of Biotechnology, Government of India, and MLP0008 & OLP0101 from CSIR-CDRI, Lucknow to SKG. SS, SP, SK, and RKS avail JRF fellowship from CSIR (Council of Scientific and Industrial Research), Government of India. RK avails SRF fellowship from ICMR (Indian Council of Medical Research), and AG and ADC avail JRF fellowship from UGC (University Grant Commission), Government of India.
Author information
Authors and Affiliations
Contributions
SKG has developed the concept, designed the study, planned experiments, analyzed results, and prepared the manuscript. SS designed the study, performed most experiments, analyzed the results, and drafted the manuscript. AG, RK, SP, SK, ADC, and PP helped with neonatal rat cardiomyocyte isolation, lentivirus, and AAV production. RKS and KM performed electron microscopy experiments on heart samples. PP and KJ helped with echocardiography and animal experiments. PB and KH provided the rat TAC heart samples. PC and SK performed the RNA sequencing analysis. TT and HH gave critical inputs and provided cardiac fractionation data.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Informed consent
TT has filed and licensed patents about non-coding RNAs and is the founder and shareholder of Cardior Pharmaceuticals GmbH (outside of this manuscript). SKG holds patents about non-coding RNAs (outside of this manuscript).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the topical collection “Mitochondria at the heart of cardioprotection”.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, S., Gaur, A., Sharma, R.K. et al. Musashi-2 causes cardiac hypertrophy and heart failure by inducing mitochondrial dysfunction through destabilizing Cluh and Smyd1 mRNA. Basic Res Cardiol 118, 46 (2023). https://doi.org/10.1007/s00395-023-01016-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00395-023-01016-y