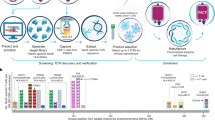
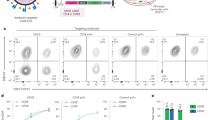
Abstract
The reliance on viral vectors for the production of genetically engineered immune cells for adoptive cellular therapies remains a translational bottleneck. Here we report a method leveraging the DNA repair pathway homology-mediated end joining, as well as optimized reagent composition and delivery, for the Cas9-induced targeted integration of large DNA payloads into primary human T cells with low toxicity and at efficiencies nearing those of viral vectors (targeted knock-in of 1–6.7 kb payloads at rates of up to 70% at multiple targeted genomic loci and with cell viabilities of over 80%). We used the method to produce T cells with an engineered T-cell receptor or a chimaeric antigen receptor and show that the cells maintained low levels of exhaustion markers and excellent capacities for proliferation and cytokine production and that they elicited potent antitumour cytotoxicity in vitro and in mice. The method is readily adaptable to current good manufacturing practices and scale-up processes, and hence may be used as an alternative to viral vectors for the production of genetically engineered T cells for cancer immunotherapies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The NGS sequencing data are available from the NCBI’s repository with the biosample accession numbers SAMN37790969, SAMN37790970, SAMN37790971, SAMN37790972, SAMN37790973, SAMN37790974, SAMN37790975, SAMN37790976, SAMN37790977 and SAMN37790978. The raw and analysed datasets generated during the study are available for research purposes from the corresponding authors on reasonable request. Source data are provided with this paper.
References
Khan, S. et al. Role of recombinant DNA technology to improve life. Int. J. Genomics Proteomics 2016, 2405954 (2016).
Spolski, R., Li, P. & Leonard, W. J. Biology and regulation of IL-2: from molecular mechanisms to human therapy. Nat. Rev. Immunol. 18, 648–659 (2018).
Maetzig, T., Galla, M., Baum, C. & Schambach, A. Gammaretroviral vectors: biology, technology and application. Viruses 3, 677–713 (2011).
Dotti, G., Gottschalk, S., Savoldo, B. & Brenner, M. K. Design and development of therapies using chimeric antigen receptor-expressing T cells. Immunol. Rev. 257, 107–126 (2014).
Walther, W. & Stein, U. Viral vectors for gene transfer: a review of their use in the treatment of human diseases. Drugs 60, 249–271 (2000).
Bulcha, J. T., Wang, Y., Ma, H., Tai, P. W. L. & Gao, G. Viral vector platforms within the gene therapy landscape. Signal Transduct. Target Ther. 6, 53 (2021).
Soundara Rajan, T., Gugliandolo, A., Bramanti, P. & Mazzon, E. In vitro-transcribed mRNA chimeric antigen receptor T cell (IVT mRNA CAR T) therapy in hematologic and solid tumor management: a preclinical update. Int. J. Mol. Sci. 21, 6514 (2020).
Kebriaei, P. et al. Phase I trials using Sleeping Beauty to generate CD19-specific CAR T cells. J. Clin. Invest. 126, 3363–3376 (2016).
Monjezi, R. et al. Enhanced CAR T-cell engineering using non-viral Sleeping Beauty transposition from minicircle vectors. Leukemia 31, 186–194 (2017).
Li, X. et al. A resurrected mammalian hAT transposable element and a closely related insect element are highly active in human cell culture. Proc. Natl Acad. Sci. USA 110, E478–E487 (2013).
Bishop, D. C. et al. PiggyBac-engineered T cells expressing CD19-specific CARs that lack IgG1 Fc spacers have potent activity against B-ALL xenografts. Mol. Ther. 26, 1883–1895 (2018).
Nguyen, D. N. et al. Polymer-stabilized Cas9 nanoparticles and modified repair templates increase genome editing efficiency. Nat. Biotechnol. 38, 44–49 (2020).
Schober, K. et al. Orthotopic replacement of T-cell receptor α- and β-chains with preservation of near-physiological T-cell function. Nat. Biomed. Eng. 3, 974–984 (2019).
Roth, T. L. et al. Reprogramming human T cell function and specificity with non-viral genome targeting. Nature 559, 405–409 (2018).
Capecchi, M. R. The new mouse genetics: altering the genome by gene targeting. Trends Genet. 5, 70–76 (1989).
Puchta, H., Dujon, B. & Hohn, B. Homologous recombination in plant cells is enhanced by in vivo induction of double strand breaks into DNA by a site-specific endonuclease. Nucleic Acids Res. 21, 5034–5040 (1993).
Anzalone, A. V., Koblan, L. W. & Liu, D. R. Genome editing with CRISPR–Cas nucleases, base editors, transposases and prime editors. Nat. Biotechnol. 38, 824–844 (2020).
Lee, J., Chung, J.-H., Kim, H. M., Kim, D.-W. & Kim, H. Designed nucleases for targeted genome editing. Plant Biotechnol. J. 14, 448–462 (2016).
Hendel, A. et al. Chemically modified guide RNAs enhance CRISPR–Cas genome editing in human primary cells. Nat. Biotechnol. 33, 985–989 (2015).
Webber, B. R. et al. Highly efficient multiplex human T cell engineering without double-strand breaks using Cas9 base editors. Nat. Commun. 10, 5222 (2019).
Johnson, M. J., Laoharawee, K., Lahr, W. S., Webber, B. R. & Moriarity, B. S. Engineering of primary human B cells with CRISPR/Cas9 targeted nuclease. Sci. Rep. 8, 12144 (2018).
Osborn, M. J. et al. Evaluation of TCR gene editing achieved by TALENs, CRISPR/Cas9, and megaTAL nucleases. Mol. Ther. 24, 570–581 (2016).
Zeng, J. et al. Therapeutic base editing of human hematopoietic stem cells. Nat. Med. 26, 535–541 (2020).
Bak, R. O., Dever, D. P. & Porteus, M. H. CRISPR/Cas9 genome editing in human hematopoietic stem cells. Nat. Protoc. 13, 358–376 (2018).
June, C. H., Blazar, B. R. & Riley, J. L. Engineering lymphocyte subsets: tools, trials and tribulations. Nat. Rev. Immunol. 9, 704–716 (2009).
Sather, B. D. et al. Efficient modification of CCR5 in primary human hematopoietic cells using a megaTAL nuclease and AAV donor template. Sci. Transl. Med. 7, 307ra156 (2015).
Osborn, M. J. et al. CRISPR/Cas9-based cellular engineering for targeted gene overexpression. Int. J. Mol. Sci. 19, 946 (2018).
Pomeroy, E. J. et al. A genetically engineered primary human natural killer cell platform for cancer immunotherapy. Mol. Ther. 28, 52–63 (2020).
Wang, D., Tai, P. W. L. & Gao, G. Adeno-associated virus vector as a platform for gene therapy delivery. Nat. Rev. Drug Discov. 18, 358–378 (2019).
Samulski, R. J. & Muzyczka, N. AAV-mediated gene therapy for research and therapeutic purposes. Annu Rev. Virol. 1, 427–451 (2014).
Deyle, D. R., Li, L. B., Ren, G. & Russell, D. W. The effects of polymorphisms on human gene targeting. Nucleic Acids Res. 42, 3119–3124 (2014).
Ang, J. X. D. et al. Considerations for homology-based DNA repair in mosquitoes: impact of sequence heterology and donor template source. PLoS Genet. 18, e1010060 (2022).
Kan, Y., Ruis, B., Lin, S. & Hendrickson, E. A. The mechanism of gene targeting in human somatic cells. PLoS Genet. 10, e1004251 (2014).
Rosenberg, S. A. & Restifo, N. P. Adoptive cell transfer as personalized immunotherapy for human cancer. Science 348, 62–68 (2015).
Chandran, S. S. & Klebanoff, C. A. T cell receptor-based cancer immunotherapy: emerging efficacy and pathways of resistance. Immunol. Rev. 290, 127–147 (2019).
Semenova, N. et al. Multiple cytosolic DNA sensors bind plasmid DNA after transfection. Nucleic Acids Res. 47, 10235–10246 (2019).
Maurisse, R. et al. Comparative transfection of DNA into primary and transformed mammalian cells from different lineages. BMC Biotechnol. 10, 9 (2010).
Chen, Q., Sun, L. & Chen, Z. J. Regulation and function of the cGAS-STING pathway of cytosolic DNA sensing. Nat. Immunol. 17, 1142–1149 (2016).
Paludan, S. R. & Bowie, A. G. Immune sensing of DNA. Immunity 38, 870–880 (2013).
Wu, J. & Chen, Z. J. Innate immune sensing and signaling of cytosolic nucleic acids. Annu. Rev. Immunol. 32, 461–488 (2014).
Clark, K., Plater, L., Peggie, M. & Cohen, P. Use of the pharmacological inhibitor BX795 to study the regulation and physiological roles of TBK1 and IkappaB kinase epsilon: a distinct upstream kinase mediates Ser-172 phosphorylation and activation. J. Biol. Chem. 284, 14136–14146 (2009).
Richters, A. et al. Identification and further development of potent TBK1 inhibitors. ACS Chem. Biol. 10, 289–298 (2015).
Decout, A., Katz, J. D., Venkatraman, S. & Ablasser, A. The cGAS–STING pathway as a therapeutic target in inflammatory diseases. Nat. Rev. Immunol. 21, 548–569 (2021).
Zhang, Z., Qiu, S., Zhang, X. & Chen, W. Optimized DNA electroporation for primary human T cell engineering. BMC Biotechnol. 18, 4 (2018).
Kay, M. A., He, C.-Y. & Chen, Z.-Y. A robust system for production of minicircle DNA vectors. Nat. Biotechnol. 28, 1287–1289 (2010).
Obst, R. The timing of T cell priming and cycling. Front. Immunol. 6, 563 (2015).
Yu, L. & Liu, P. Cytosolic DNA sensing by cGAS: regulation, function, and human diseases. Signal Transduct. Target Ther. 6, 170 (2021).
Vance, R. E. Cytosolic DNA sensing: the field narrows. Immunity 45, 227–228 (2016).
Zahid, A., Ismail, H., Li, B. & Jin, T. Molecular and structural basis of DNA sensors in antiviral innate immunity. Front. Immunol. 11, 613039 (2020).
Wierson, W. A. et al. Efficient targeted integration directed by short homology in zebrafish and mammalian cells. Elife 9, e53968 (2020).
Xue, C. & Greene, E. C. DNA repair pathway choices in CRISPR–Cas9-mediated genome editing. Trends Genet. 37, 639–656 (2021).
Li, X. et al. Efficient SSA-mediated precise genome editing using CRISPR/Cas9. FEBS J. 285, 3362–3375 (2018).
Bewg, W. P., Ci, D. & Tsai, C.-J. Genome editing in trees: from multiple repair pathways to long-term stability. Front. Plant Sci. 9, 1732 (2018).
Terada, K. et al. Isolation of TCR genes with tumor-killing activity from tumor-infiltrating and circulating lymphocytes in a tumor rejection cynomolgus macaque model. Mol. Ther. Oncolytics 24, 77–86 (2022).
de Vree, P. J. P. et al. Targeted sequencing by proximity ligation for comprehensive variant detection and local haplotyping. Nat. Biotechnol. 32, 1019–1025 (2014).
Yao, X. et al. Homology-mediated end joining-based targeted integration using CRISPR/Cas9. Cell Res. 27, 801–814 (2017).
Dahlman, J. E. et al. Orthogonal gene knockout and activation with a catalytically active Cas9 nuclease. Nat. Biotechnol. 33, 1159–1161 (2015).
Philip, B. et al. A highly compact epitope-based marker/suicide gene for easier and safer T-cell therapy. Blood 124, 1277–1287 (2014).
Berger, A. Th1 and Th2 responses: what are they? Brit. Med. J. 321, 424 (2000).
Eyquem, J. et al. Targeting a CAR to the TRAC locus with CRISPR/Cas9 enhances tumour rejection. Nature 543, 113–117 (2017).
DeRenzo, C. & Gottschalk, S. Genetic modification strategies to enhance CAR T cell persistence for patients with solid tumors. Front. Immunol. 10, 218 (2019).
Liu, X. et al. A chimeric switch–receptor targeting PD1 augments the efficacy of second-generation CAR T cells in advanced solid tumors. Cancer Res. 76, 1578–1590 (2016).
Yeku, O. O., Purdon, T. J., Koneru, M., Spriggs, D. & Brentjens, R. J. Armored CAR T cells enhance antitumor efficacy and overcome the tumor microenvironment. Sci. Rep. 7, 10541 (2017).
Yu, S., Yi, M., Qin, S. & Wu, K. Next generation chimeric antigen receptor T cells: safety strategies to overcome toxicity. Mol. Cancer 18, 125 (2019).
Brandt, L. J. B., Barnkob, M. B., Michaels, Y. S., Heiselberg, J. & Barington, T. Emerging approaches for regulation and control of CAR T cells: a mini review. Front. Immunol. 11, 326 (2020).
Srivastava, S. et al. Logic-gated ROR1 chimeric antigen receptor expression rescues T cell-mediated toxicity to normal tissues and enables selective tumor targeting. Cancer Cell 35, 489–503 (2019).
Raes, L., De Smedt, S. C., Raemdonck, K. & Braeckmans, K. Non-viral transfection technologies for next-generation therapeutic T cell engineering. Biotechnol. Adv. 49, 107760 (2021).
Odé, Z., Condori, J., Peterson, N., Zhou, S. & Krenciute, G. CRISPR-mediated non-viral site-specific gene integration and expression in T cells: protocol and application for T-cell therapy. Cancers 12, 1704 (2020).
Pearce, E. L., Poffenberger, M. C., Chang, C.-H. & Jones, R. G. Fueling immunity: insights into metabolism and lymphocyte function. Science 342, 1242454 (2013).
Paludan, S. R., Reinert, L. S. & Hornung, V. DNA-stimulated cell death: implications for host defence, inflammatory diseases and cancer. Nat. Rev. Immunol. 19, 141–153 (2019).
Ceccaldi, R., Rondinelli, B. & D’Andrea, A. D. Repair pathway choices and consequences at the double-strand break. Trends Cell Biol. 26, 52–64 (2016).
Suzuki, K. et al. In vivo genome editing via CRISPR/Cas9 mediated homology-independent targeted integration. Nature 540, 144–149 (2016).
Lock, D. et al. Automated, scaled, transposon-based production of CAR T cells. J. Immunother. Cancer 10, e005189 (2022).
Schweizer, M. & Merten, O.-W. Large-scale production means for the manufacturing of lentiviral vectors. Curr. Gene Ther. 10, 474–486 (2010).
Merten, O.-W., Hebben, M. & Bovolenta, C. Production of lentiviral vectors. Mol. Ther. Methods Clin. Dev. 3, 16017 (2016).
Kaiser, A. D. et al. Towards a commercial process for the manufacture of genetically modified T cells for therapy. Cancer Gene Ther. 22, 72–78 (2015).
Wang, X. & Rivière, I. Clinical manufacturing of CAR T cells: foundation of a promising therapy. Mol. Ther. Oncolytics 3, 16015 (2016).
Gee, A. P. GMP CAR-T cell production. Best Pract. Res. Clin. Haematol. 31, 126–134 (2018).
Alves, C. P. A., Prazeres, D. M. F. & Monteiro, G. A. Minicircle biopharmaceuticals—an overview of purification strategies. Front. Chem. Eng. 2, (2021).
Tian, Y., Li, Y., Shao, Y. & Zhang, Y. Gene modification strategies for next-generation CAR T cells against solid cancers. J. Hematol. Oncol. 13, 54 (2020).
Nakamura, K. et al. Autologous antigen-presenting cells efficiently expand piggyBac transposon CAR-T cells with predominant memory phenotype. Mol. Ther. Methods Clin. Dev. 21, 315–324 (2021).
Bishop, D. C. et al. Development of CAR T-cell lymphoma in 2 of 10 patients effectively treated with piggyBac-Modified CD19 CAR T cells. Blood 138, 1504–1509 (2021).
Lukjanov, V., Koutná, I. & Šimara, P. CAR T-cell production using nonviral approaches. J. Immunol. Res. 2021, 6644685 (2021).
Chicaybam, L. et al. Transposon-mediated generation of CAR-T cells shows efficient anti B-cell leukemia response after ex vivo expansion. Gene Ther. 27, 85–95 (2020).
Kuleshov, M. V. et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 44, W90–W97 (2016).
Tsukahara, T. et al. CD19 target-engineered T-cells accumulate at tumor lesions in human B-cell lymphoma xenograft mouse models. Biochem. Biophys. Res. Commun. 438, 84–89 (2013).
Hu, S.-I. et al. Pre-clinical assessment of chimeric antigen receptor t cell therapy targeting CD19+ B cell malignancy. Ann. Transl. Med. 8, 584 (2020).
Acknowledgements
We thank J. Garbie and the University of Minnesota Genomic Core for their assistance throughout the project. J.G.S. is supported by the T32HL007062-46 Hematology Research Training Program. B.S.M. is supported by the following NIH grants (CA136393, CA254849, AI161017, and AI161017). B.R.W. received support from NIH AI163731, CA237789, CA276345, the Minnesota Ovarian Cancer Alliance in 2021 and the Randy Shaver Cancer Research and Community Fund in 2022.
Author information
Authors and Affiliations
Contributions
B.R.W., M.J.J., J.G.S., N.J.S., T.H., M.C., R.S.M. and B.S.M. conceived and planned the experiments. B.R.W., M.J.J., J.G.S., N.J.S., W.S.L., A.P.D., X.Q., B.R., M.D.D. and B.W. carried out the experiments. B.R.W., M.J.J., J.G.S., T.K.S., L.J.M. and B.S.M. analysed data and interpreted the results. B.R.W., M.J.J., J.G.S. and B.S.M. wrote the manuscript. All authors provided critical feedback during the study and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
B.R.W., R.S.M. and B.S.M. are principal investigators of Sponsored Research Agreements funded by Intima Biosciences to support this work. Patents have been filed covering the methods and approaches outlined in this work.
Peer review
Peer review information
Nature Biomedical Engineering thanks Zoltán Ivics, Zsuzsanna Izsvak, Matthew Porteus and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Longer homology arms increase integration with both HMEJ or HR.
Percentage of cells expressing GFP following electroporation with Cas9 mRNA, gRNAs, and HMEJ templates with the indicated homology arm lengths. Statistical analyses were done using Two-way ANOVA (n = 3–6 independent biological donors) (*p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001).
Extended Data Fig. 2 Lentivirally transduced cells have higher expression levels of CD19 CAR than non-viral engineered cells.
MFI of samples following HMEJ non-viral genome engineering or lentiviral transduction with a construct encoding CD19CAR-2A-RQR8.
Extended Data Fig. 3 HMEJ-engineered CD19 CAR T cells express similar levels of activation and exhaustion surface markers as lentivirally transduced cells.
(A) Expression of 41BB, CD25, CD69, and Ox40 in RQR8+CD4+, total CD4+, RQR8+CD8+ and total CD8+ T cell subsets following HMEJ non-viral genome engineering or lentiviral transduction with a construct encoding CD19CAR-2A-RQR8. (B) Expression of Lag3, PD1, and TIM3 in RQR8+CD4+, total CD4+, RQR8+CD8+ and total CD8+ T cell subsets following HMEJ non-viral genome engineering or lentiviral transduction with a construct encoding CD19CAR-2A-RQR8. Lack of statistically significant differences was determined by comparing TRAC, AAVS1, and pulse-only cells to Lenti cells using One-way ANOVA followed by Dunnett’s multiple comparison test.
Extended Data Fig. 4 HMEJ-engineered and lentivirally transduced CD19 CAR T cells produce cytokines in response to target cells, as measured by ICS.
Percentage of cells expressing cytokines IFNγ, TNF, and IL2 as well as a degranulation marker CD107a in CD4 (left panels) and CD8 (right panels) CD19 CAR T cells following coculture with CD19+ Raji target cells.
Extended Data Fig. 5 HMEJ-engineered and lentivirally transduced CD19 CAR T cells produce cytokines in response to target cells, as measured by Luminex.
Concentration of IFNγ, TNF, IL4, and IL5 in the supernatant of CD19 CAR T cells following co-culture with CD19+ Raji target cells. All statistical analyses were done using One-way ANOVA followed by Tukey’s multiple comparison test. (n = 6 independent biological donors) (*p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001).
Supplementary information
Supplementary Information
Supplementary figures and tables.
Supplementary data
Source data for the supplementary figures.
Source data
Source Data for Figs. 1–6 and Extended Data Figs. 1–5
Source data and statistics.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Webber, B.R., Johnson, M.J., Skeate, J.G. et al. Cas9-induced targeted integration of large DNA payloads in primary human T cells via homology-mediated end-joining DNA repair. Nat. Biomed. Eng (2023). https://doi.org/10.1038/s41551-023-01157-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41551-023-01157-4