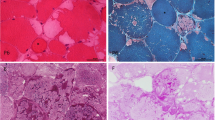
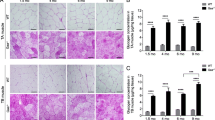
Abstract
At least five enzymes including three E3 ubiquitin ligases are dedicated to glycogen’s spherical structure. Absence of any reverts glycogen to a structure resembling amylopectin of the plant kingdom. This amylopectinosis (polyglucosan body formation) causes fatal neurological diseases including adult polyglucosan body disease (APBD) due to glycogen branching enzyme deficiency, Lafora disease (LD) due to deficiencies of the laforin glycogen phosphatase or the malin E3 ubiquitin ligase and type 1 polyglucosan body myopathy (PGBM1) due to RBCK1 E3 ubiquitin ligase deficiency. Little is known about these enzymes’ functions in glycogen structuring. Toward understanding these functions, we undertake a comparative murine study of the amylopectinoses of APBD, LD and PGBM1. We discover that in skeletal muscle, polyglucosan bodies form as two main types, small and multitudinous (‘pebbles’) or giant and single (‘boulders’), and that this is primarily determined by the myofiber types in which they form, ‘pebbles’ in glycolytic and ‘boulders’ in oxidative fibers. This pattern recapitulates what is known in the brain in LD, innumerable dust-like in astrocytes and single giant sized in neurons. We also show that oxidative myofibers are relatively protected against amylopectinosis, in part through highly increased glycogen branching enzyme expression. We present evidence of polyglucosan body size-dependent cell necrosis. We show that sex influences amylopectinosis in genotype, brain region and myofiber-type-specific fashion. RBCK1 is a component of the linear ubiquitin chain assembly complex (LUBAC), the only known cellular machinery for head-to-tail linear ubiquitination critical to numerous cellular pathways. We show that the amylopectinosis of RBCK1 deficiency is not due to loss of linear ubiquitination, and that another function of RBCK1 or LUBAC must exist and operate in the shaping of glycogen. This work opens multiple new avenues toward understanding the structural determinants of the mammalian carbohydrate reservoir critical to neurologic and neuromuscular function and disease.










Similar content being viewed by others
Data availability
Data are available in the main text. All raw data are available upon request.
References
Aboujaoude A, Minassian B, Mitra S (2021) LUBAC: a new player in polyglucosan body disease. Biochem Soc Trans 49:2443–2454. https://doi.org/10.1042/BST20210838
Aguado C, Sarkar S, Korolchuk VI, Criado O, Vernia S, Boya P et al (2010) Laforin, the most common protein mutated in Lafora disease, regulates autophagy. Hum Mol Genet 19:2867–2876. https://doi.org/10.1093/hmg/ddq190
Ahonen S, Nitschke S, Grossman TR, Kordasiewicz H, Wang P, Zhao X et al (2021) Gys1 antisense therapy rescues neuropathological bases of murine Lafora disease. Brain 144:2985–2993. https://doi.org/10.1093/brain/awab194
Akman HO, Sheiko T, Tay SK, Finegold MJ, Dimauro S, Craigen WJ (2011) Generation of a novel mouse model that recapitulates early and adult onset glycogenosis type IV. Hum Mol Genet 20:4430–4439. https://doi.org/10.1093/hmg/ddr371
Auge E, Duran J, Guinovart JJ, Pelegri C, Vilaplana J (2018) Exploring the elusive composition of corpora amylacea of human brain. Sci Rep 8:13525. https://doi.org/10.1038/s41598-018-31766-y
Auge E, Pelegri C, Manich G, Cabezon I, Guinovart JJ, Duran J et al (2018) Astrocytes and neurons produce distinct types of polyglucosan bodies in Lafora disease. Glia 66:2094–2107. https://doi.org/10.1002/glia.23463
Augusto VPRC, Campos ERG (2004) Skeletal muscle fiber types in C57BL6J mice. Braz J morphol sci 21:89–94
Bak LK, Schousboe A, Waagepetersen HS (2006) The glutamate/GABA-glutamine cycle: aspects of transport, neurotransmitter homeostasis and ammonia transfer. J Neurochem 98:641–653. https://doi.org/10.1111/j.1471-4159.2006.03913.x
Baumann RJ, Kocoshis SA, Wilson D (1983) Lafora disease: liver histopathology in presymptomatic children. Ann Neurol 14:86–89. https://doi.org/10.1002/ana.410140116
Berard-Badier M, Pellissier JF, Gambarelli D, de Barsy T, Roger J, Toga M (1980) The retina in Lafora disease: light and electron microscopy. Albrecht Von Graefes Arch Klin Exp Ophthalmol 212:285–294. https://doi.org/10.1007/BF00410522
Bijman J, Quinton PM (1987) Lactate and bicarbonate uptake in the sweat duct of cystic fibrosis and normal subjects. Pediatr Res 21:79–82. https://doi.org/10.1203/00006450-198701000-00017
Boisson B, Laplantine E, Dobbs K, Cobat A, Tarantino N, Hazen M et al (2015) Human HOIP and LUBAC deficiency underlies autoinflammation, immunodeficiency, amylopectinosis, and lymphangiectasia. J Exp Med 212:939–951. https://doi.org/10.1084/jem.20141130
Brooks GA (1986) The lactate shuttle during exercise and recovery. Med Sci Sports Exerc 18:360–368. https://doi.org/10.1249/00005768-198606000-00019
Brown AM, Baltan Tekkok S, Ransom BR (2004) Energy transfer from astrocytes to axons: the role of CNS glycogen. Neurochem Int 45:529–536. https://doi.org/10.1016/j.neuint.2003.11.005
Bruno C, Servidei S, Shanske S, Karpati G, Carpenter S, McKee D et al (1993) Glycogen branching enzyme deficiency in adult polyglucosan body disease. Ann Neurol 33:88–93. https://doi.org/10.1002/ana.410330114
Eyster HC (1959) The optimum pH for diastase of malt activity. Ohio J Sci 59:257–262
Carpenter S, Karpati G (1981) Sweat gland duct cells in Lafora disease: diagnosis by skin biopsy. Neurology 31:1564–1568. https://doi.org/10.1212/wnl.31.12.1564
Carpenter S, Karpati G, Andermann F, Jacob JC, Andermann E (1974) Lafora’s disease: peroxisomal storage in skeletal muscle. Neurology 24:531–538. https://doi.org/10.1212/wnl.24.6.531
Cenacchi G, Papa V, Costa R, Pegoraro V, Marozzo R, Fanin M et al (2019) Update on polyglucosan storage diseases. Virchows Arch 475:671–686. https://doi.org/10.1007/s00428-019-02633-6
Cenci U, Nitschke F, Steup M, Minassian BA, Colleoni C, Ball SG (2014) Transition from glycogen to starch metabolism in Archaeplastida. Trends Plant Sci 19:18–28. https://doi.org/10.1016/j.tplants.2013.08.004
Chambers JK, Thongtharb A, Shiga T, Azakami D, Saito M, Sato M et al (2018) Accumulation of laforin and other related proteins in canine Lafora disease with EPM2B repeat expansion. Vet Pathol 55:543–551. https://doi.org/10.1177/0300985818758471
Chan EM, Young EJ, Ianzano L, Munteanu I, Zhao X, Christopoulos CC et al (2003) Mutations in NHLRC1 cause progressive myoclonus epilepsy. Nat Genet 35:125–127. https://doi.org/10.1038/ng1238
Chen L, Wang N, Hu W, Yu X, Yang R, Han Y et al (2021) Polyglucosan body myopathy 1 may cause cognitive impairment: a case report from China. BMC Musculoskelet Disord 22:35. https://doi.org/10.1186/s12891-020-03884-0
Chown EE, Wang P, Zhao X, Crowder JJ, Strober JW, Sullivan MA et al (2020) GYS1 or PPP1R3C deficiency rescues murine adult polyglucosan body disease. Ann Clin Transl Neurol 7:2186–2198. https://doi.org/10.1002/acn3.51211
Cohen P, Kelsall IR, Nanda SK, Zhang J (2020) HOIL-1, an atypical E3 ligase that controls MyD88 signalling by forming ester bonds between ubiquitin and components of the Myddosome. Adv Biol Regul 75:100666. https://doi.org/10.1016/j.jbior.2019.100666
Coleman DL, Gambetti P, Mauro SD, Blume RE (1974) Muscle in Lafora disease. Arch Neurol 31:396–406. https://doi.org/10.1001/archneur.1974.00490420062007
Criado O, Aguado C, Gayarre J, Duran-Trio L, Garcia-Cabrero AM, Vernia S et al (2012) Lafora bodies and neurological defects in malin-deficient mice correlate with impaired autophagy. Hum Mol Genet 21:1521–1533. https://doi.org/10.1093/hmg/ddr590
Dainese L, Monin ML, Demeret S, Brochier G, Froissart R, Spraul A et al (2013) Abnormal glycogen in astrocytes is sufficient to cause adult polyglucosan body disease. Gene 515:376–379. https://doi.org/10.1016/j.gene.2012.12.065
Delgado-Garcia JM (2001) Structure and function of the cerebellum. Rev Neurol 33:635–642
DePaoli-Roach AA, Tagliabracci VS, Segvich DM, Meyer CM, Irimia JM, Roach PJ (2010) Genetic depletion of the malin E3 ubiquitin ligase in mice leads to lafora bodies and the accumulation of insoluble laforin. J Biol Chem 285:25372–25381. https://doi.org/10.1074/jbc.M110.148668
Dimauro I, Antonioni A, Mercatelli N, Grazioli E, Fantini C, Barone R et al (2019) The early response of alphaB-crystallin to a single bout of aerobic exercise in mouse skeletal muscles depends upon fiber oxidative features. Redox Biol 24:101183. https://doi.org/10.1016/j.redox.2019.101183
Dringen R, Gebhardt R, Hamprecht B (1993) Glycogen in astrocytes: possible function as lactate supply for neighboring cells. Brain Res 623:208–214. https://doi.org/10.1016/0006-8993(93)91429-v
Dubowitz V, Sewry AC, Oldfors A (2020) Muscle biopsy: A practical approach. Elsevier, London
Duran J, Hervera A, Markussen KH, Varea O, Lopez-Soldado I, Sun RC et al (2021) Astrocytic glycogen accumulation drives the pathophysiology of neurodegeneration in Lafora disease. Brain 144:2349–2360. https://doi.org/10.1093/brain/awab110
Elton L, Carpentier I, Verhelst K, Staal J, Beyaert R (2015) The multifaceted role of the E3 ubiquitin ligase HOIL-1: beyond linear ubiquitination. Immunol Rev 266:208–221. https://doi.org/10.1111/imr.12307
Feher J (2017) Muscle Energetics, Fatigue, and Training. Quantitative Human Physiology, 2nd edn. Academic Press, Cambridge, pp 334–348
Fujita H, Tokunaga A, Shimizu S, Whiting AL, Aguilar-Alonso F, Takagi K et al (2018) Cooperative domain formation by homologous motifs in HOIL-1L and SHARPIN plays a crucial role in LUBAC stabilization. Cell Rep 23:1192–1204. https://doi.org/10.1016/j.celrep.2018.03.112
Fuseya Y, Fujita H, Kim M, Ohtake F, Nishide A, Sasaki K et al (2020) The HOIL-1L ligase modulates immune signalling and cell death via monoubiquitination of LUBAC. Nat Cell Biol 22:663–673. https://doi.org/10.1038/s41556-020-0517-9
Fuseya Y, Iwai K (2021) Biochemistry, pathophysiology, and regulation of linear ubiquitination: intricate regulation by coordinated functions of the associated ligase and deubiquitinase. Cells. https://doi.org/10.3390/cells10102706
Ganesh S, Delgado-Escueta AV, Sakamoto T, Avila MR, Machado-Salas J, Hoshii Y et al (2002) Targeted disruption of the Epm2a gene causes formation of Lafora inclusion bodies, neurodegeneration, ataxia, myoclonus epilepsy and impaired behavioral response in mice. Hum Mol Genet 11:1251–1262. https://doi.org/10.1093/hmg/11.11.1251
Gentry MS, Worby CA, Dixon JE (2005) Insights into Lafora disease: malin is an E3 ubiquitin ligase that ubiquitinates and promotes the degradation of laforin. Proc Natl Acad Sci U S A 102:8501–8506. https://doi.org/10.1073/pnas.0503285102
Goebel HH, Shin YS, Gullotta F, Yokota T, Alroy J, Voit T et al (1992) Adult polyglucosan body myopathy. J Neuropathol Exp Neurol 51:24–35. https://doi.org/10.1097/00005072-199201000-00004
Goldsmith E, Sprang S, Fletterick R (1982) Structure of maltoheptaose by difference Fourier methods and a model for glycogen. J Mol Biol 156:411–427. https://doi.org/10.1016/0022-2836(82)90336-9
Gray F, Gherardi R, Marshall A, Janota I, Poirier J (1988) Adult polyglucosan body disease (APBD). J Neuropathol Exp Neurol 47:459–474. https://doi.org/10.1097/00005072-198807000-00007
Gumusgoz E, Guisso DR, Kasiri S, Wu J, Dear M, Verhalen B et al (2021) Targeting Gys1 with AAV-SaCas9 decreases pathogenic polyglucosan bodies and neuroinflammation in adult polyglucosan body and Lafora disease mouse models. Neurotherapeutics 18:1414–1425. https://doi.org/10.1007/s13311-021-01040-7
Gumusgoz E, Kasiri S, Guisso DR, Wu J, Dear M, Verhalen B et al (2022) AAV-mediated artificial miRNA reduces pathogenic polyglucosan bodies and neuroinflammation in adult polyglucosan body and Lafora disease mouse models. Neurotherapeutics 19:982–993. https://doi.org/10.1007/s13311-022-01218-7
Harriman DG, Millar JH, Stevenson AC (1955) Progressive familial myoclonic epilepsy in three families: its clinical features and pathological basis. Brain 78:325–349. https://doi.org/10.1093/brain/78.3.325
Hedberg-Oldfors C, Oldfors A (2015) Polyglucosan storage myopathies. Mol Aspects Med 46:85–100. https://doi.org/10.1016/j.mam.2015.08.006
Heitkotter H, Linderman RE, Cava JA, Woertz EN, Mastey RR, Summerfelt P et al (2021) Retinal alterations in patients with Lafora disease. Am J Ophthalmol Case Rep 23:101146. https://doi.org/10.1016/j.ajoc.2021.101146
Hrdinka M, Gyrd-Hansen M (2017) The Met1-linked ubiquitin machinery: emerging themes of (de)regulation. Mol Cell 68:265–280. https://doi.org/10.1016/j.molcel.2017.09.001
Jungermann K, Kietzmann T (1996) Zonation of parenchymal and nonparenchymal metabolism in liver. Annu Rev Nutr 16:179–203. https://doi.org/10.1146/annurev.nu.16.070196.001143
Kanow MA, Giarmarco MM, Jankowski CS, Tsantilas K, Engel AL, Du J et al (2017) Biochemical adaptations of the retina and retinal pigment epithelium support a metabolic ecosystem in the vertebrate eye. Elife. https://doi.org/10.7554/eLife.28899
Kaufman MA, Dwork AJ, Willson NJ, John S, Liu JD (1993) Late-onset Lafora’s disease with typical intraneuronal inclusions. Neurology 43:1246–1248. https://doi.org/10.1212/wnl.43.6.1246
Kelsall IR, McCrory EH, Xu Y, Scudamore CL, Nanda SK, Mancebo-Gamella P et al (2022) HOIL-1 ubiquitin ligase activity targets unbranched glucosaccharides and is required to prevent polyglucosan accumulation. EMBO J 41:e109700. https://doi.org/10.15252/embj.2021109700
Kelsall IR, Zhang J, Knebel A, Arthur JSC, Cohen P (2019) The E3 ligase HOIL-1 catalyses ester bond formation between ubiquitin and components of the Myddosome in mammalian cells. Proc Natl Acad Sci U S A 116:13293–13298. https://doi.org/10.1073/pnas.1905873116
Kilimann MW, Oldfors A (2015) Glycogen pathways in disease: new developments in a classical field of medical genetics. J Inherit Metab Dis 38:483–487. https://doi.org/10.1007/s10545-014-9785-5
Knecht E, Criado-Garcia O, Aguado C, Gayarre J, Duran-Trio L, Garcia-Cabrero AM et al (2012) Malin knockout mice support a primary role of autophagy in the pathogenesis of Lafora disease. Autophagy 8:701–703. https://doi.org/10.4161/auto.19522
Kobayashi K, Iyoda K, Ohtsuka Y, Ohtahara S, Yamada M (1990) Longitudinal clinicoelectrophysiologic study of a case of Lafora disease proven by skin biopsy. Epilepsia 31:194–201. https://doi.org/10.1111/j.1528-1167.1990.tb06306.x
Lafora GRG, B. (1911) Beitrag zur histopathogie der myoklonischen epilepsie [German]. Z Gesamte Neurol Psychiatr 6:1–14
Lahuerta M, Gonzalez D, Aguado C, Fathinajafabadi A, Garcia-Gimenez JL, Moreno-Estelles M et al (2020) Reactive glia-derived neuroinflammation: a novel hallmark in Lafora progressive myoclonus epilepsy that progresses with age. Mol Neurobiol 57:1607–1621. https://doi.org/10.1007/s12035-019-01842-z
Lechtenberg BC, Rajput A, Sanishvili R, Dobaczewska MK, Ware CF, Mace PD et al (2016) Structure of a HOIP/E2~ubiquitin complex reveals RBR E3 ligase mechanism and regulation. Nature 529:546–550. https://doi.org/10.1038/nature16511
Lerin C, Montell E, Berman HK, Newgard CB, Gomez-Foix AM (2000) Overexpression of protein targeting to glycogen in cultured human muscle cells stimulates glycogen synthesis independent of glycogen and glucose 6-phosphate levels. J Biol Chem 275:39991–39995. https://doi.org/10.1074/jbc.M006251200
Lopez-Gonzalez I, Viana R, Sanz P, Ferrer I (2017) Inflammation in Lafora disease: evolution with disease progression in laforin and malin knock-out mouse models. Mol Neurobiol 54:3119–3130. https://doi.org/10.1007/s12035-016-9884-4
Lossos A, Meiner Z, Barash V, Soffer D, Schlesinger I, Abramsky O et al (1998) Adult polyglucosan body disease in Ashkenazi Jewish patients carrying the Tyr329Ser mutation in the glycogen-branching enzyme gene. Ann Neurol 44:867–872. https://doi.org/10.1002/ana.410440604
Magistretti PJ (2011) Neuron-glia metabolic coupling and plasticity. Exp Physiol 96:407–410. https://doi.org/10.1113/expphysiol.2010.053157
Magistretti PJ, Morrison JH (1988) Noradrenaline and vasoactive intestinal peptide-containing neuronal systems in neocortex: functional convergence with contrasting morphology. Neuroscience 24:367–378. https://doi.org/10.1016/0306-4522(88)90338-7
Magistretti PJ, Pellerin L, Rothman DL, Shulman RG (1999) Energy on demand. Science 283:496–497. https://doi.org/10.1126/science.283.5401.496
Maile CA, Hingst JR, Mahalingan KK, O’Reilly AO, Cleasby ME, Mickelson JR et al (2017) A highly prevalent equine glycogen storage disease is explained by constitutive activation of a mutant glycogen synthase. Biochim Biophys Acta Gen Subj 1861:3388–3398. https://doi.org/10.1016/j.bbagen.2016.08.021
Manchester J, Skurat AV, Roach P, Hauschka SD, Lawrence JC Jr (1996) Increased glycogen accumulation in transgenic mice overexpressing glycogen synthase in skeletal muscle. Proc Natl Acad Sci U S A 93:10707–10711. https://doi.org/10.1073/pnas.93.20.10707
Minassian BA, Lee JR, Herbrick JA, Huizenga J, Soder S, Mungall AJ et al (1998) Mutations in a gene encoding a novel protein tyrosine phosphatase cause progressive myoclonus epilepsy. Nat Genet 20:171–174. https://doi.org/10.1038/2470
Mitra S, Chen B, Wang P, Chown EE, Dear M, Guisso DR et al (2023) Laforin targets malin to glycogen in Lafora progressive myoclonus epilepsy. Dis Model Mech. https://doi.org/10.1242/dmm.049802
Murphy RM, Xu H, Latchman H, Larkins NT, Gooley PR, Stapleton DI (2012) Single fiber analyses of glycogen-related proteins reveal their differential association with glycogen in rat skeletal muscle. Am J Physiol Cell Physiol 303:C1146-1155. https://doi.org/10.1152/ajpcell.00252.2012
Neville HE, Brooke MH, Austin JH (1974) Studies in myoclonus epilepsy. (Lafora body form). IV Skeletal muscle abnormalities Arch Neurol 30:466–474. https://doi.org/10.1001/archneur.1974.00490360042009
Nilsson J, Schoser B, Laforet P, Kalev O, Lindberg C, Romero NB et al (2013) Polyglucosan body myopathy caused by defective ubiquitin ligase RBCK1. Ann Neurol 74:914–919. https://doi.org/10.1002/ana.23963
Nitschke F, Sullivan MA, Wang P, Zhao X, Chown EE, Perri AM et al (2017) Abnormal glycogen chain length pattern, not hyperphosphorylation, is critical in Lafora disease. EMBO Mol Med 9:906–917. https://doi.org/10.15252/emmm.201707608
Nitschke S, Sullivan MA, Mitra S, Marchioni CR, Lee JPY, Smith BH et al (2022) Glycogen synthase downregulation rescues the amylopectinosis of murine RBCK1 deficiency. Brain 145:2361–2377. https://doi.org/10.1093/brain/awac017
Oda H, Beck DB, Kuehn HS, Sampaio Moura N, Hoffmann P, Ibarra M et al (2019) Second case of HOIP deficiency expands clinical features and defines inflammatory transcriptome regulated by LUBAC. Front Immunol 10:479. https://doi.org/10.3389/fimmu.2019.00479
Orhan Akman H, Emmanuele V, Kurt YG, Kurt B, Sheiko T, DiMauro S et al (2015) A novel mouse model that recapitulates adult-onset glycogenosis type 4. Hum Mol Genet 24:6801–6810. https://doi.org/10.1093/hmg/ddv385
Pellerin L, Magistretti PJ (1994) Glutamate uptake into astrocytes stimulates aerobic glycolysis: a mechanism coupling neuronal activity to glucose utilization. Proc Natl Acad Sci U S A 91:10625–10629. https://doi.org/10.1073/pnas.91.22.10625
Peltzer N, Darding M, Montinaro A, Draber P, Draberova H, Kupka S et al (2018) LUBAC is essential for embryogenesis by preventing cell death and enabling haematopoiesis. Nature 557:112–117. https://doi.org/10.1038/s41586-018-0064-8
Peltzer N, Rieser E, Taraborrelli L, Draber P, Darding M, Pernaute B et al (2014) HOIP deficiency causes embryonic lethality by aberrant TNFR1-mediated endothelial cell death. Cell Rep 9:153–165. https://doi.org/10.1016/j.celrep.2014.08.066
Phadke R, Hedberg-Oldfors C, Scalco RS, Lowe DM, Ashworth M, Novelli M et al (2020) RBCK1-related disease: a rare multisystem disorder with polyglucosan storage, auto-inflammation, recurrent infections, skeletal, and cardiac myopathy-Four additional patients and a review of the current literature. J Inherit Metab Dis 43:1002–1013. https://doi.org/10.1002/jimd.12234
Potter CS, Wang Z, Silva KA, Kennedy VE, Stearns TM, Burzenski L et al (2014) Chronic proliferative dermatitis in Sharpin null mice: development of an autoinflammatory disease in the absence of B and T lymphocytes and IL4/IL13 signaling. PLoS ONE 9:e85666. https://doi.org/10.1371/journal.pone.0085666
Puri R, Suzuki T, Yamakawa K, Ganesh S (2012) Dysfunctions in endosomal-lysosomal and autophagy pathways underlie neuropathology in a mouse model for Lafora disease. Hum Mol Genet 21:175–184. https://doi.org/10.1093/hmg/ddr452
Raben N, Danon M, Lu N, Lee E, Shliselfeld L, Skurat AV et al (2001) Surprises of genetic engineering: a possible model of polyglucosan body disease. Neurology 56:1739–1745. https://doi.org/10.1212/wnl.56.12.1739
Reusche E, Aksu F, Goebel HH, Shin YS, Yokota T, Reichmann H (1992) A mild juvenile variant of type IV glycogenosis. Brain Dev 14:36–43. https://doi.org/10.1016/s0387-7604(12)80277-4
Rich LR, Harris W, Brown AM (2019) The role of brain glycogen in supporting physiological function. Front Neurosci 13:1176. https://doi.org/10.3389/fnins.2019.01176
Roach PJ, Depaoli-Roach AA, Hurley TD, Tagliabracci VS (2012) Glycogen and its metabolism: some new developments and old themes. Biochem J 441:763–787. https://doi.org/10.1042/BJ20111416
Roach PJ, Skurat AV, Harris RA (2001) Regulation of Glycogen Metabolism. Handbook of physiology, vol II. Lippincott Company, Philadelphia, pp 609–647
Robitaille Y, Carpenter S, Karpati G, DiMauro SD (1980) A distinct form of adult polyglucosan body disease with massive involvement of central and peripheral neuronal processes and astrocytes: a report of four cases and a review of the occurrence of polyglucosan bodies in other conditions such as Lafora’s disease and normal ageing. Brain 103:315–336. https://doi.org/10.1093/brain/103.2.315
Rodriguez Carvajal A, Grishkovskaya I, Gomez Diaz C, Vogel A, Sonn-Segev A, Kushwah MS et al (2021) The linear ubiquitin chain assembly complex (LUBAC) generates heterotypic ubiquitin chains. Elife 10(10):e60660. https://doi.org/10.7554/eLife.60660
Rubio-Villena C, Viana R, Bonet J, Garcia-Gimeno MA, Casado M, Heredia M et al (2018) Astrocytes: new players in progressive myoclonus epilepsy of Lafora type. Hum Mol Genet 27:1290–1300. https://doi.org/10.1093/hmg/ddy044
Sanchez-Martin P, Lahuerta M, Viana R, Knecht E, Sanz P (2020) Regulation of the autophagic PI3KC3 complex by laforin/malin E3-ubiquitin ligase, two proteins involved in Lafora disease. Biochim Biophys Acta Mol Cell Res 1867:118613. https://doi.org/10.1016/j.bbamcr.2019.118613
Sathiah P, Gochhait D, Dehuri P, Subramanian H (2017) Diagnosis of Lafora disease by skin biopsy. J Clin Diagn Res. https://doi.org/10.7860/JCDR/2017/28570.10552
Schiaffino S, Reggiani C (2011) Fiber types in mammalian skeletal muscles. Physiol Rev 91:1447–1531. https://doi.org/10.1152/physrev.00031.2010
Schwarz GA, Yanoff M (1965) Lafora’s Disease. Distinct clinico-pathologic form of Unverricht’s syndrome. Arch Neurol 12:172–188. https://doi.org/10.1001/archneur.1965.00460260062008
Scott W, Stevens J, Binder-Macleod SA (2001) Human skeletal muscle fiber type classifications. Phys Ther 81:1810–1816
Servidei S, Riepe RE, Langston C, Tani LY, Bricker JT, Crisp-Lindgren N et al (1987) Severe cardiopathy in branching enzyme deficiency. J Pediatr 111:51–56. https://doi.org/10.1016/s0022-3476(87)80341-4
Sheehan DC, Hrapchak BB (1980) Theory and practice of histotechnology. Mosby, Maryland Heights
Smit JJ, Monteferrario D, Noordermeer SM, van Dijk WJ, van der Reijden BA, Sixma TK (2012) The E3 ligase HOIP specifies linear ubiquitin chain assembly through its RING-IBR-RING domain and the unique LDD extension. EMBO J 31:3833–3844. https://doi.org/10.1038/emboj.2012.217
Smit JJ, van Dijk WJ, El Atmioui D, Merkx R, Ovaa H, Sixma TK (2013) Target specificity of the E3 ligase LUBAC for ubiquitin and NEMO relies on different minimal requirements. J Biol Chem 288:31728–31737. https://doi.org/10.1074/jbc.M113.495846
Stieglitz B, Morris-Davies AC, Koliopoulos MG, Christodoulou E, Rittinger K (2012) LUBAC synthesizes linear ubiquitin chains via a thioester intermediate. EMBO Rep 13:840–846. https://doi.org/10.1038/embor.2012.105
Stieglitz B, Rana RR, Koliopoulos MG, Morris-Davies AC, Schaeffer V, Christodoulou E et al (2013) Structural basis for ligase-specific conjugation of linear ubiquitin chains by HOIP. Nature 503:422–426. https://doi.org/10.1038/nature12638
Sullivan MA, Nitschke S, Skwara EP, Wang P, Zhao X, Pan XS et al (2019) Skeletal muscle glycogen chain length correlates with insolubility in mouse models of polyglucosan-associated neurodegenerative diseases. Cell Rep 27(1334–1344):e1336. https://doi.org/10.1016/j.celrep.2019.04.017
Sun RC, Young LEA, Bruntz RC, Markussen KH, Zhou Z, Conroy LR et al (2021) Brain glycogen serves as a critical glucosamine cache required for protein glycosylation. Cell Metab. https://doi.org/10.1016/j.cmet.2021.05.003
Minagawa T (1943) Purified diastases: properties of diastase action. PJAB 19:553–556
Tagliabracci VS, Girard JM, Segvich D, Meyer C, Turnbull J, Zhao X et al (2008) Abnormal metabolism of glycogen phosphate as a cause for Lafora disease. J Biol Chem 283:33816–33825. https://doi.org/10.1074/jbc.M807428200
Tagliabracci VS, Turnbull J, Wang W, Girard JM, Zhao X, Skurat AV et al (2007) Laforin is a glycogen phosphatase, deficiency of which leads to elevated phosphorylation of glycogen in vivo. Proc Natl Acad Sci U S A 104:19262–19266. https://doi.org/10.1073/pnas.0707952104
Tang B, Frasinyuk MS, Chikwana VM, Mahalingan KK, Morgan CA, Segvich DM et al (2020) Discovery and development of small-molecule inhibitors of glycogen synthase. J Med Chem 63:3538–3551. https://doi.org/10.1021/acs.jmedchem.9b01851
Thomsen C, Malfatti E, Jovanovic A, Roberts M, Kalev O, Lindberg C et al (2022) Proteomic characterisation of polyglucosan bodies in skeletal muscle in RBCK1 deficiency. Neuropathol Appl Neurobiol. https://doi.org/10.1111/nan.12761
Tokunaga F, Iwai K (2012) Linear ubiquitination: a novel NF-kappaB regulatory mechanism for inflammatory and immune responses by the LUBAC ubiquitin ligase complex. Endocr J 59:641–652. https://doi.org/10.1507/endocrj.ej12-0148
Tokunaga F, Sakata S, Saeki Y, Satomi Y, Kirisako T, Kamei K et al (2009) Involvement of linear polyubiquitylation of NEMO in NF-kappaB activation. Nat Cell Biol 11:123–132. https://doi.org/10.1038/ncb1821
Turnbull J, Epp JR, Goldsmith D, Zhao X, Pencea N, Wang P et al (2014) PTG protein depletion rescues malin-deficient Lafora disease in mouse. Ann Neurol 75:442–446. https://doi.org/10.1002/ana.24104
Turnbull J, Girard JM, Lohi H, Chan EM, Wang P, Tiberia E et al (2012) Early-onset Lafora body disease. Brain 135:2684–2698. https://doi.org/10.1093/brain/aws205
Turnbull J, Girard JM, Pencea N, Zhao X, Graham TE, Wang P et al (2011) Lafora bodies in skeletal muscle are fiber type specific. Neurology 76:1674–1676. https://doi.org/10.1212/WNL.0b013e318219faf6
Turnbull J, Tiberia E, Striano P, Genton P, Carpenter S, Ackerley CA et al (2016) Lafora disease. Epileptic Disord 18:38–62. https://doi.org/10.1684/epd.2016.0842
Turnbull J, Wang P, Girard JM, Ruggieri A, Wang TJ, Draginov AG et al (2010) Glycogen hyperphosphorylation underlies lafora body formation. Ann Neurol 68:925–933. https://doi.org/10.1002/ana.22156
Valles-Ortega J, Duran J, Garcia-Rocha M, Bosch C, Saez I, Pujadas L et al (2011) Neurodegeneration and functional impairments associated with glycogen synthase accumulation in a mouse model of Lafora disease. EMBO Mol Med 3:667–681. https://doi.org/10.1002/emmm.201100174
Van Hoof F, Hageman-Bal M (1967) Progressive familial myoclonic epilepsy with Lafora bodies. Electron microscopic and histochemical study of a cerebral biopsy. Acta Neuropathol 7:315–336. https://doi.org/10.1007/BF00688087
Varea O, Duran J, Aguilera M, Prats N, Guinovart JJ (2021) Suppression of glycogen synthesis as a treatment for Lafora disease: Establishing the window of opportunity. Neurobiol Dis 147:105173. https://doi.org/10.1016/j.nbd.2020.105173
Verhalen B, Arnold S, Minassian BA (2018) Lafora Disease: a review of molecular mechanisms and pathology. Neuropediatrics 49:357–362. https://doi.org/10.1055/s-0038-1675238
Vilchez D, Ros S, Cifuentes D, Pujadas L, Valles J, Garcia-Fojeda B et al (2007) Mechanism suppressing glycogen synthesis in neurons and its demise in progressive myoclonus epilepsy. Nat Neurosci 10:1407–1413. https://doi.org/10.1038/nn1998
Vohra R, Aldana BI, Bulli G, Skytt DM, Waagepetersen H, Bergersen LH et al (2019) Lactate-mediated protection of retinal ganglion cells. J Mol Biol 431:1878–1888. https://doi.org/10.1016/j.jmb.2019.03.005
Woods AE, Roy C (1994) Laboratory histopathology: a complete reference. Churchill Livingstone Press, London
Worby CA, Gentry MS, Dixon JE (2006) Laforin, a dual specificity phosphatase that dephosphorylates complex carbohydrates. J Biol Chem 281:30412–30418. https://doi.org/10.1074/jbc.M606117200
Yamanami S, Ishihara T, Takahashi M, Uchino F (1992) Comparative study of intraneuronal polyglucosan bodies in brains from patients with Lafora disease and aged dogs. Acta Pathol Jpn 42:787–792. https://doi.org/10.1111/j.1440-1827.1992.tb01879.x
Yoshimura N (2013) Brain pathology of Lafora Disease: localization of polyglucosan aggrgations (Lafora bodies) within neuronal networks of cerebrellar cortex. J Hirosaki Univ Health Welfare 4:13–24
Acknowledgements
We thank UT Southwestern’s Histopathology, Quantitative Light Microscopy (supported by NIH grant P30CA1425431) and Whole Brain Microscopy Cores and Children’s Health’s Electron Microscopy Core, especially Ricardo Olivarez, Marcel Mettlen and Alejandra Vargas.
Funding
The work was funded by NIH grant (P01NS097197 to BM), Japan Society for Promotion of Science KAKENHI grant (22H04988) to KI and UT Southwestern Medical Center Wellstone Cooperative Research Center grant to SM. BM, SM and FN are also supported by Chan-Zuckerberg Initiative Patient-Partnered Collaborations for Rare Neurodegenerative Disease grant (2022-316703).
Author information
Authors and Affiliations
Contributions
Conceptualization: SM, BAM. Investigation: SM, BC, JMS, SN, FN LC, MD, TL, MV, JW, YF. Validation: SM, BAM. Formal analysis: SM, BME, BAM. Resources: KI, FN, BME, BAM. Writing original draft: SM, BAM. Writing, review and editing: SM, BME, BAM. Supervision: SM, BAM. Project administration: BAM. Funding acquisition: SM, KI, BAM.
Corresponding authors
Ethics declarations
Conflict of interests
The authors declare that they do not have any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mitra, S., Chen, B., Shelton, J.M. et al. Myofiber-type-dependent ‘boulder’ or ‘multitudinous pebble’ formations across distinct amylopectinoses. Acta Neuropathol 147, 46 (2024). https://doi.org/10.1007/s00401-024-02698-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00401-024-02698-x