Abstract
Claudin 18.2, a tight-junction molecule predominantly found in the nonmalignant gastric epithelium, becomes accessible on the tumour cell surface during malignant transformation, thereby providing an appealing target for cancer therapy. Data from two phase III trials testing the anti-claudin 18.2 antibody zolbetuximab have established claudin 18.2-positive advanced-stage gastric cancers as an independent therapeutic subset that derives benefit from the addition of this agent to chemotherapy. This development has substantially increased the percentage of patients eligible for targeted therapy. Furthermore, newer treatments, such as high-affinity monoclonal antibodies, bispecific antibodies, chimeric antigen receptor T cells and antibody–drug conjugates capable of bystander killing effects, have shown considerable promise in patients with claudin 18.2-expressing gastric cancers. This new development has resulted from drug developers moving beyond traditional targets, such as driver gene alterations or growth factors. In this Review, we highlight the biological rationale and explore the clinical activity of therapies that target claudin 18.2 in patients with advanced-stage gastric cancer and explore the potential for expansion of claudin 18.2-targeted therapies to patients with other claudin 18.2-positive solid tumours.
Key points
-
Claudin 18.2 is expressed almost exclusively in the gastric mucosa, and no clear evidence exists of a role of this tight-junction protein in the carcinogenesis and/or proliferation of gastric cancer.
-
Two pivotal phase III trials to test zolbetuximab, a monoclonal antibody that targets claudin 18.2, have demonstrated statistically significant improvements in both the progression-free and overall survival of patients with unresectable gastric cancer and have established claudin 18.2 as a validated therapeutic target.
-
Determination of the optimal treatment sequence, especially for patients who are potentially eligible for several targeted therapies or immunotherapies, as well as the feasibility of biomarker tests for multiple proteins, will be the subject of much debate following the clinical implementation of zolbetuximab.
-
Claudin 18.2 can potentially be targeted using a wide range of therapeutic modalities beyond monoclonal antibodies including bispecific antibodies, antibody–drug conjugates, chimeric antigen receptor T cells and mRNA-based approaches.
-
The development of claudin 18.2-targeted therapies is expanding and will probably encompass other claudin 18.2-positive cancer types.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Smyth, E. C., Nilsson, M., Grabsch, H. I., van Grieken, N. C. & Lordick, F. Gastric cancer. Lancet 396, 635–648 (2020).
Kang, Y. K. et al. Nivolumab plus chemotherapy versus placebo plus chemotherapy in patients with HER2-negative, untreated, unresectable advanced or recurrent gastric or gastro-oesophageal junction cancer (ATTRACTION-4): a randomised, multicentre, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 23, 234–247 (2022).
Janjigian, Y. Y. et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet 398, 27–40 (2021).
Lordick, F. et al. Gastric cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 33, 1005–1020 (2022).
Nagatsuma, A. K. et al. Expression profiles of HER2, EGFR, MET and FGFR2 in a large cohort of patients with gastric adenocarcinoma. Gastric Cancer 18, 227–238 (2015).
Nakamura, Y., Kawazoe, A., Lordick, F., Janjigian, Y. Y. & Shitara, K. Biomarker-targeted therapies for advanced-stage gastric and gastro-oesophageal junction cancers: an emerging paradigm. Nat. Rev. Clin. Oncol. 18, 473–487 (2021).
Shitara, K. et al. Nivolumab plus chemotherapy or ipilimumab in gastro-oesophageal cancer. Nature 603, 942–948 (2022).
Yeong, J. et al. Choice of PD-L1 immunohistochemistry assay influences clinical eligibility for gastric cancer immunotherapy. Gastric Cancer 25, 741–750 (2022).
Shitara, K. et al. Zolbetuximab plus mFOLFOX6 in patients with CLDN18.2-positive, HER2-negative, untreated, locally advanced unresectable or metastatic gastric or gastro-oesophageal junction adenocarcinoma (SPOTLIGHT): a multicentre, randomised, double-blind, phase 3 trial. Lancet 401, 1655–1668 (2023).
Shah, M. A. et al. Zolbetuximab plus CAPOX in CLDN18.2-positive gastric or gastroesophageal junction adenocarcinoma: the randomized, phase 3 GLOW trial. Nat. Med. 29, 2133–2141 (2023).
Qi, C. et al. Claudin18.2-specific CAR T cells in gastrointestinal cancers: phase 1 trial interim results. Nat. Med. 28, 1189–1198 (2022).
Janjigian, Y. Y. et al. A multi-cohort phase I/IIa clinical trial to evaluate the safety, tolerability, and pharmacokinetics of TST001 administered as a monotherapy, with nivolumab or standard of care in patients with locally advanced or metastatic solid tumors: TransStar101. J. Clin. Oncol. 41, (Suppl. 16), Abstr. TPS4176 (2023).
Xu, R.-H. et al. A phase 1a dose-escalation, multicenter trial of anti-claudin 18.2 antibody drug conjugate CMG901 in patients with resistant/refractory solid tumors. J. Clin. Oncol. 41, (Suppl. 4), Abstr. 352 (2023).
Barbier, A. J., Jiang, A. Y., Zhang, P., Wooster, R. & Anderson, D. G. The clinical progress of mRNA vaccines and immunotherapies. Nat. Biotechnol. 40, 840–854 (2022).
Sahin, U. et al. Claudin-18 splice variant 2 is a pan-cancer target suitable for therapeutic antibody development. Clin. Cancer Res. 14, 7624–7634 (2008).
Tsukita, S., Itoh, M., Nagafuchi, A., Yonemura, S. & Tsukita, S. Submembranous junctional plaque proteins include potential tumor suppressor molecules. J. Cell Biol. 123, 1049–1053 (1993).
Zihni, C., Mills, C., Matter, K. & Balda, M. S. Tight junctions: from simple barriers to multifunctional molecular gates. Nat. Rev. Mol. Cell Biol. 17, 564–580 (2016).
Schneeberger, E. E. & Lynch, R. D. Structure, function, and regulation of cellular tight junctions. Am. J. Physiol. 262, L647–L661 (1992).
Gunzel, D. & Yu, A. S. Claudins and the modulation of tight junction permeability. Physiol. Rev. 93, 525–569 (2013).
Gumbiner, B. M. Breaking through the tight junction barrier. J. Cell Biol. 123, 1631–1633 (1993).
Anderson, J. M. & Van Itallie, C. M. Tight junctions and the molecular basis for regulation of paracellular permeability. Am. J. Physiol. 269, G467–G475 (1995).
Staehelin, L. A. Further observations on the fine structure of freeze-cleaved tight junctions. J. Cell Sci. 13, 763–786 (1973).
Tsukita, S. & Furuse, M. Pores in the wall: claudins constitute tight junction strands containing aqueous pores. J. Cell Biol. 149, 13–16 (2000).
Furuse, M., Fujita, K., Hiiragi, T., Fujimoto, K. & Tsukita, S. Claudin-1 and -2: novel integral membrane proteins localizing at tight junctions with no sequence similarity to occludin. J. Cell Biol. 141, 1539–1550 (1998).
Morin, P. J. Claudin proteins in human cancer: promising new targets for diagnosis and therapy. Cancer Res. 65, 9603–9606 (2005).
Niimi, T. et al. Claudin-18, a novel downstream target gene for the T/EBP/NKX2.1 homeodomain transcription factor, encodes lung- and stomach-specific isoforms through alternative splicing. Mol. Cell Biol. 21, 7380–7390 (2001).
Türeci, O. et al. Claudin-18 gene structure, regulation, and expression is evolutionary conserved in mammals. Gene 481, 83–92 (2011).
Sahin, U. et al. FAST: a randomised phase II study of zolbetuximab (IMAB362) plus EOX versus EOX alone for first-line treatment of advanced CLDN18.2-positive gastric and gastro-oesophageal adenocarcinoma. Ann. Oncol. 32, 609–619 (2021).
Jovov, B. et al. Claudin-18: a dominant tight junction protein in Barrett’s esophagus and likely contributor to its acid resistance. Am. J. Physiol. Gastrointest. Liver Physiol. 293, G1106–G1113 (2007).
Hayashi, D. et al. Deficiency of claudin-18 causes paracellular H+ leakage, up-regulation of interleukin-1beta, and atrophic gastritis in mice. Gastroenterology 142, 292–304 (2012).
Hewitt, K. J., Agarwal, R. & Morin, P. J. The claudin gene family: expression in normal and neoplastic tissues. BMC Cancer 6, 186 (2006).
Jun, K. H., Kim, J. H., Jung, J. H., Choi, H. J. & Chin, H. M. Expression of claudin-7 and loss of claudin-18 correlate with poor prognosis in gastric cancer. Int. J. Surg. 12, 156–162 (2014).
Chen, X. et al. Clinicopathological significance of claudin 4 expression in gastric carcinoma: a systematic review and meta-analysis. Oncol. Targets Ther. 27, 3205–3212 (2016).
Park, J. Y. et al. Up-regulated claudin 7 expression in intestinal-type gastric carcinoma. Oncol. Rep. 18, 377–382 (2007).
Kohmoto, T. et al. Claudin-6 is a single prognostic marker and functions as a tumor-promoting gene in a subgroup of intestinal type gastric cancer. Gastric Cancer 23, 403–417 (2020).
Hedrick, L., Cho, K. R. & Vogelstein, B. Cell adhesion molecules as tumour suppressors. Trends Cell Biol. 3, 36–39 (1993).
Resnick, M. B., Konkin, T., Routhier, J., Sabo, E. & Pricolo, V. E. Claudin-1 is a strong prognostic indicator in stage II colonic cancer: a tissue microarray study. Mod. Pathol. 18, 511–518 (2005).
Al Moustafa, A. E. et al. Identification of genes associated with head and neck carcinogenesis by cDNA microarray comparison between matched primary normal epithelial and squamous carcinoma cells. Oncogene 21, 2634–2640 (2002).
Sanada, Y. et al. Down-regulation of the claudin-18 gene, identified through serial analysis of gene expression data analysis, in gastric cancer with an intestinal phenotype. J. Pathol. 208, 633–642 (2006).
Matsuda, Y. et al. Gastric and intestinal claudin expression at the invasive front of gastric carcinoma. Cancer Sci. 98, 1014–1019 (2007).
Dottermusch, M., Kruger, S., Behrens, H. M., Halske, C. & Rocken, C. Expression of the potential therapeutic target claudin-18.2 is frequently decreased in gastric cancer: results from a large Caucasian cohort study. Virchows Arch. 475, 563–571 (2019).
Oshima, T. et al. Down-regulation of claudin-18 is associated with the proliferative and invasive potential of gastric cancer at the invasive front. PLoS ONE 8, e74757 (2013).
Zhang, S. J. et al. miR-1303 targets claudin-18 gene to modulate proliferation and invasion of gastric cancer cells. Dig. Dis. Sci. 59, 1754–1763 (2014).
Suzuki, K. et al. Deficiency of stomach-type claudin-18 in mice induces gastric tumor formation independent of H. pylori infection. Cell Mol. Gastroenterol. Hepatol. 8, 119–142 (2019).
Swisshelm, K., Macek, R. & Kubbies, M. Role of claudins in tumorigenesis. Adv. Drug Deliv. Rev. 57, 919–928 (2005).
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature 513, 202–209 (2014).
Zhang, W. H. et al. The significance of the CLDN18-ARHGAP fusion gene in gastric cancer: a systematic review and meta-analysis. Front. Oncol. 10, 1214 (2020).
Nakayama, I. et al. Enrichment of CLDN18-ARHGAP fusion gene in gastric cancers in young adults. Cancer Sci. 110, 1352–1363 (2019).
Kataoka, K. & Ogawa, S. Variegated RHOA mutations in human cancers. Exp. Hematol. 44, 1123–1129 (2016).
Yang, H. et al. RhoGAP domain-containing fusions and PPAPDC1A fusions are recurrent and prognostic in diffuse gastric cancer. Nat. Commun. 9, 4439 (2018).
Komatsu, M. et al. ARHGAP-RhoA signaling provokes homotypic adhesion-triggered cell death of metastasized diffuse-type gastric cancer. Oncogene 41, 4779–4794 (2022).
Tanaka, M. et al. Claudin-18 is an early-stage marker of pancreatic carcinogenesis. J. Histochem. Cytochem. 59, 942–952 (2011).
Sanada, Y. et al. Immunohistochemical study of claudin 18 involvement in intestinal differentiation during the progression of intraductal papillary mucinous neoplasm. Anticancer Res. 30, 2995–3003 (2010).
Lee, J. H. et al. Immunohistochemical analysis of claudin expression in pancreatic cystic tumors. Oncol. Rep. 25, 971–978 (2011).
Shinozaki, A. et al. Claudin-18 in biliary neoplasms. Its significance in the classification of intrahepatic cholangiocarcinoma. Virchows Arch. 459, 73–80 (2011).
Matsuda, M. et al. Immunohistochemical analysis of colorectal cancer with gastric phenotype: claudin-18 is associated with poor prognosis. Pathol. Int. 60, 673–680 (2010).
Woll, S. et al. Claudin 18.2 is a target for IMAB362 antibody in pancreatic neoplasms. Int. J. Cancer 134, 731–739 (2014).
Micke, P. et al. Aberrantly activated claudin 6 and 18.2 as potential therapy targets in non-small-cell lung cancer. Int. J. Cancer 135, 2206–2214 (2014).
Keira, Y. et al. An immunohistochemical marker panel including claudin-18, maspin, and p53 improves diagnostic accuracy of bile duct neoplasms in surgical and presurgical biopsy specimens. Virchows Arch. 466, 265–277 (2015).
Takasawa, K. et al. Claudin-18 coupled with EGFR/ERK signaling contributes to the malignant potentials of bile duct cancer. Cancer Lett. 403, 66–73 (2017).
Iwaya, M. et al. Colitis-associated colorectal adenocarcinomas frequently express claudin 18 isoform 2: implications for claudin 18.2 monoclonal antibody therapy. Histopathology 79, 227–237 (2021).
Zhang, Z., Liu, X., Zhou, L., Zhang, M. & Liang, Z. Investigation of clinical application of claudin 18 isoform 2 in pancreatic ductal adenocarcinoma: a retrospective analysis of 302 Chinese patients. Histol. Histopathol. 37, 1031–1040 (2022).
Arpa, G. et al. Claudin-18 expression in small bowel adenocarcinoma: a clinico-pathologic study. Virchows Arch. 481, 853–863 (2022).
Wang, X. et al. Claudin 18.2 is a potential therapeutic target for zolbetuximab in pancreatic ductal adenocarcinoma. World J. Gastrointest. Oncol. 14, 1252–1264 (2022).
Wang, F. et al. Claudin18.2 as a potential therapeutic target for primary ovarian mucinous carcinomas and metastatic ovarian mucinous carcinomas from upper gastrointestinal primary tumours. BMC Cancer 23, 44 (2023).
Kayikcioglu, E. & Yuceer, R. O. The role of claudin 18.2 and HER-2 in pancreatic cancer outcomes. Medicine 102, e32882 (2023).
Park, S. et al. Clinicopathological features and prognosis of resected pancreatic ductal adenocarcinoma patients with claudin-18 overexpression. J. Clin. Med. 12, 5394 (2023).
Yano, K., Imaeda, T. & Niimi, T. Transcriptional activation of the human claudin-18 gene promoter through two AP-1 motifs in PMA-stimulated MKN45 gastric cancer cells. Am. J. Physiol. Gastrointest. Liver Physiol. 294, G336–G343 (2008).
Ito, T. et al. Transcriptional regulation of claudin-18 via specific protein kinase C signaling pathways and modification of DNA methylation in human pancreatic cancer cells. J. Cell Biochem. 112, 1761–1772 (2011).
Chen, J. et al. Targeting CLDN18.2 in cancers of the gastrointestinal tract: new drugs and new indications. Front. Oncol. 13, 1132319 (2023).
Moentenich, V. et al. Claudin 18.2 expression in esophageal adenocarcinoma and its potential impact on future treatment strategies. Oncol. Lett. 19, 3665–3670 (2020).
Karanjawala, Z. E. et al. New markers of pancreatic cancer identified through differential gene expression analyses: claudin 18 and annexin A8. Am. J. Surg. Pathol. 32, 188–196 (2008).
Sanada, Y. et al. Histopathologic evaluation of stepwise progression of pancreatic carcinoma with immunohistochemical analysis of gastric epithelial transcription factor SOX2: comparison of expression patterns between invasive components and cancerous or nonneoplastic intraductal components. Pancreas 32, 164–170 (2006).
Halimi, S. A. et al. Claudin-18 overexpression in intestinal-type mucinous borderline tumour of the ovary. Histopathology 63, 534–544 (2013).
Halimi, S. A. et al. Comprehensive immunohistochemical analysis of the gastrointestinal and Mullerian phenotypes of 139 ovarian mucinous cystadenomas. Hum. Pathol. 109, 21–30 (2021).
Kubota, Y. et al. Comprehensive clinical and molecular characterization of claudin 18.2 expression in advanced gastric or gastroesophageal junction cancer. ESMO Open 8, 100762 (2023).
Kayikcioglu, E., Yuceer, R. O., Cetin, B., Yuceer, K. & Karahan, N. Prognostic value of claudin 18.2 expression in gastric adenocarcinoma. World J. Gastrointest. Oncol. 15, 343–351 (2023).
Jia, K. et al. Multiplex immunohistochemistry defines the tumor immune microenvironment and immunotherapeutic outcome in CLDN18.2-positive gastric cancer. BMC Med. 20, 223 (2022).
Arnold, A. et al. Prognostic impact of claudin 18.2 in gastric and esophageal adenocarcinomas. Clin. Transl. Oncol. 22, 2357–2363 (2020).
Kim, S. R. et al. Clinical significance of CLDN18.2 expression in metastatic diffuse-type gastric cancer. J. Gastric Cancer 20, 408–420 (2020).
Hong, J. Y. et al. Claudin 18.2 expression in various tumor types and its role as a potential target in advanced gastric cancer. Transl. Cancer Res. 9, 3367–3374 (2020).
Rohde, C. et al. Comparison of claudin 18.2 expression in primary tumors and lymph node metastases in Japanese patients with gastric adenocarcinoma. Jpn J. Clin. Oncol. 49, 870–876 (2019).
Baek, J. H. et al. Clinical implications of claudin18.2 expression in patients with gastric cancer. Anticancer Res. 39, 6973–6979 (2019).
Coati, I. et al. Claudin-18 expression in oesophagogastric adenocarcinomas: a tissue microarray study of 523 molecularly profiled cases. Br. J. Cancer 121, 257–263 (2019).
Ungureanu, B. S. et al. Clinicopathologic relevance of claudin 18.2 expression in gastric cancer: a meta-analysis. Front. Oncol. 11, 643872 (2021).
Moran, D., Maurus, D., Rohde, C. & Arozullah, A. 103P. Prevalence of CLDN18.2, HER2 and PD-L1 in gastric cancer samples. Ann. Oncol. 29, viii32 (2018).
Shitara, K. et al. Global prevalence of CLDN18.2 in patients with locally advanced (LA) unresectable or metastatic gastric or gastroesophageal junction (mG/GEJ) adenocarcinoma: biomarker analysis of two zolbetuximab phase 3 studies (SPOTLIGHT and GLOW). J. Clin. Oncol. 41, (Suppl. 16), Abstr. 4035 (2023).
Pellino, A. et al. Association of CLDN18 protein expression with clinicopathological features and prognosis in advanced gastric and gastroesophageal junction adenocarcinomas. J. Pers. Med. 11, 1095 (2021).
Xu, B. et al. Highly expressed claudin18.2 as a potential therapeutic target in advanced gastric signet-ring cell carcinoma (SRCC). J. Gastrointest. Oncol. 11, 1431–1439 (2020).
Shu, Y. et al. Prognostic significance of frequent CLDN18-ARHGAP26/6 fusion in gastric signet-ring cell cancer. Nat. Commun. 9, 2447 (2018).
Li, J. et al. Analysis of the expression and genetic alteration of CLDN18 in gastric cancer. Aging 12, 14271–14284 (2020).
Kim, S. T. et al. Comprehensive molecular characterization of gastric cancer patients from phase II second-line ramucirumab plus paclitaxel therapy trial. Genome Med. 13, 11 (2021).
Lenz, A. et al. Claudin 18 (CLDN18) gene expression and related molecular profile in gastric cancer (GC). J. Clin. Oncol. 40, (Suppl. 16), Abstr. 4048 (2022).
Shinozaki, A. et al. Epstein-Barr virus-associated gastric carcinoma: a distinct carcinoma of gastric phenotype by claudin expression profiling. J. Histochem. Cytochem. 57, 775–785 (2009).
Fukayama, M. et al. Thirty years of Epstein-Barr virus-associated gastric carcinoma. Virchows Arch. 476, 353–365 (2020).
Kadono, T. et al. The impact of claudin-18.2 expression in patients with advanced gastric cancer treated with nivolumab. J. Clin. Oncol. 41, (Suppl. 4), Abstr. 439 (2023).
Wang, Y. et al. 1230P. Immunotherapies for gastric cancer with CLDN18-ARHGAP fusion gene. Ann. Oncol. 33, S1112 (2022).
Kumagai, S. et al. An oncogenic alteration creates a microenvironment that promotes tumor progression by conferring a metabolic advantage to regulatory T cells. Immunity 53, 187–203.e188 (2020).
Shitara, K. et al. Phase 1 trial of zolbetuximab in Japanese patients with CLDN18.2+ gastric or gastroesophageal junction adenocarcinoma. Cancer Sci. 114, 1606–1615 (2023).
Lordick, F. et al. Health-related quality of life (hrqol) in patients with claudin-18 isoform 2-positive (CLDN18.2+) locally advanced (LA) unresectable or metastatic gastric or gastroesophageal junction (mg/GEJ) adenocarcinoma: results from SPOTLIGHT and GLOW. Ann. Oncol. 34, S852–S886 (2023).
Nakayama, T. et al. 2P Antitumor activity of zolbetuximab combined with chemotherapy and anti-mouse P42. PD-1 antibody (anti-mPD-1) in a syngeneic mouse model and a virtual preclinical trial using a quantitative systems pharmacology (QSP) model [abstr.]. Ann. Oncol. 33, S559–S560 (2022).
Yang, J. et al. Clinical pharmacology profile of the claudin 18.2 antibody zolbetuximab. J. Clin. Oncol. 42, (Suppl. 3), Abstr. 316 (2024).
Shen, L. et al. 1254P Updated report of a phase I study of TST001, a humanized anti-CLDN18.2 monoclonal antibody, in combination with capecitabine and oxaliplatin (CAPOX) as a first-line treatment of advanced G/GEJ cancer. Ann. Oncol. 33, S1121 (2022).
Gong, J. et al. A phase I study of TST001, a high affinity humanized anti-CLDN18.2 monoclonal antibody, in combination with capecitabine and oxaliplatin (CAPOX) as a first-line treatment of advanced G/GEJ cancer. J. Clin. Oncol. 40, (Suppl. 16) abstr. 4062 (2022).
Shen, L. et al. Osemitamab in combination with capecitabine and oxaliplatin (CAPOX) as a first line treatment of advanced G/GEJ cancer: updated data of cohort C from a phase I/IIa, multi-center study (TranStar102/TST001-1002). J. Clin. Oncol. 41, (Suppl. 16), Abstr. 4046 (2023).
Guo, W. et al. TST001 (a high affinity humanized anti-claudin18.2 monoclonal antibody) in combination with nivolumab plus capecitabine and oxaliplatin as first-line or with nivolumab as late-line treatment in locally advanced and metastatic gastric/gastroesophageal junction (G/GEJ) cancer: design of cohorts from a phase I/IIa study (TST001-1002). J. Clin. Oncol. 41, (Suppl. 4), Abstr. TPS476 (2023).
Li, J. et al. A multicenter, phase 1 study of AB011, a recombinant humanized anti-CLDN18.2 monoclonal antibody, as monotherapy and combined with capecitabine and oxaliplatin (CAPOX) in patients with advanced solid tumors. J. Clin. Oncol. 41, (Suppl. 4), Abstr. 391 (2023).
Huang, J. et al. Safety and preliminary efficacy of MIL93 in patients with advanced solid tumors: the monotherapy part of a phase 1 trial. J. Clin. Oncol. 41, (Suppl. 4), Abstr. 798 (2023).
Zhang, M. et al. A phase I/II study of ASKB589 (anti-claudin 18.2 [CLDN18.2] monoclonal antibody) in patients with solid tumors. J. Clin. Oncol. 41, (Suppl. 4), Abstr. 397 (2023).
Peng, Z. et al. A phase Ib/II study of ASKB589 (anti-claudin 18.2 [CLDN18.2] monoclonal antibody) combined with CAPOX and PD-1 inhibitor as first-line treatment for locally advanced, relapsed and metastatic gastric/gastro-esophageal junction (G/GEJ) adenocarcinoma. J. Clin. Oncol. 42, (Suppl. 3), Abstr. 317 (2024).
Konno, H. et al. ZL-1211 exhibits robust antitumor activity by enhancing ADCC and activating NK cell-mediated inflammation in CLDN18.2-high and -low expressing gastric cancer models. Cancer Res. Commun. 2, 937–950 (2022).
Sharma, S. et al. Preliminary results of a phase 1/2, first-in-human, open-label, dose escalation study of ZL-1211 (anti-Claudin 18.2 mAb) in patients with unresectable or metastatic solid tumors. J. Clin. Oncol. 41, (Suppl. 16), Abstr. 2537 (2023).
Labrijn, A. F., Janmaat, M. L., Reichert, J. M. & Parren, P. Bispecific antibodies: a mechanistic review of the pipeline. Nat. Rev. Drug Discov. 18, 585–608 (2019).
Topp, M. S. et al. Safety and activity of blinatumomab for adult patients with relapsed or refractory B-precursor acute lymphoblastic leukaemia: a multicentre, single-arm, phase 2 study. Lancet Oncol. 16, 57–66 (2015).
Kantarjian, H. et al. Blinatumomab versus chemotherapy for advanced acute lymphoblastic leukemia. N. Engl. J. Med. 376, 836–847 (2017).
Gong, J. et al. Safety results of Q-1802, a Claudin18.2/PD-L1 bsABs, in patients with relapsed or refractory solid tumors in a phase 1 study. J. Clin. Oncol. 40 (suppl 16), abstr. 2568 (2022).
Wang, Y. K. et al. Interim results of a first-in-human phase 1 study of Q-1802, a CLDN18.2/PD-L1 bsABs, in patients with relapsed or refractory solid tumors. J. Clin. Oncol. 41, (Suppl. 4), Abstr. 382 (2023).
Overman, M. J. A phase I, first-in-human, open-label, dose escalation and expansion study of PT886 in adult patients with advanced gastric, gastroesophageal junction, and pancreatic adenocarcinomas. J. Clin. Oncol. 41, (Suppl. 4), Abstr. TPS765 (2023).
Gao, J. et al. CLDN18.2 and 4-1BB bispecific antibody givastomig exerts antitumor activity through CLDN18.2-expressing tumor-directed T-cell activation. J. Immunother. Cancer 11, e006704 (2023).
Jiang, W., Wang, Z., Sheng, Z., Jung, J. & Taylor, G. 702 TJ-CD4B (ABL111), a claudin18. 2-targeted 4-1BB tumor engager induces potent tumor-dependent immune response without dose-limiting toxicity in preclinical studies. J. Immunother. Cancer 9, A730 (2021).
Hong, M., Clubb, J. D. & Chen, Y. Y. Engineering CAR-T cells for next-generation cancer therapy. Cancer Cell 38, 473–488 (2020).
Porter, D. L., Levine, B. L., Kalos, M., Bagg, A. & June, C. H. Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia. N. Engl. J. Med. 365, 725–733 (2011).
Kalos, M. et al. T cells with chimeric antigen receptors have potent antitumor effects and can establish memory in patients with advanced leukemia. Sci. Transl. Med. 3, 95ra73 (2011).
Jiang, H. et al. Claudin18.2-specific chimeric antigen receptor engineered T cells for the treatment of gastric cancer. J. Natl Cancer Inst. 111, 409–418 (2019).
Zhan, X. et al. Phase I trial of claudin 18.2-specific chimeric antigen receptor T cells for advanced gastric and pancreatic adenocarcinoma. J. Clin. Oncol. 37, (Suppl. 15), Abstr. 2509 (2019).
FDA. FDA investigating serious risk of T-cell malignancy following BCMA-directed or CD19-directed autologous chimeric antigen receptor (CAR) T cell immunotherapies. https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/fda-investigating-serious-risk-t-cell-malignancy-following-bcma-directed-or-cd19-directed-autologous (2023).
Shitara, K. et al. Trastuzumab deruxtecan in previously treated HER2-positive gastric cancer. N. Engl. J. Med. 382, 2419–2430 (2020).
Modi, S. et al. Trastuzumab deruxtecan in previously treated HER2-positive breast cancer. N. Engl. J. Med. 382, 610–621 (2020).
Cortes, J. et al. Trastuzumab deruxtecan versus trastuzumab emtansine for breast cancer. N. Engl. J. Med. 386, 1143–1154 (2022).
Chau, C. H., Steeg, P. S. & Figg, W. D. Antibody-drug conjugates for cancer. Lancet 394, 793–804 (2019).
Drago, J. Z., Modi, S. & Chandarlapaty, S. Unlocking the potential of antibody–drug conjugates for cancer therapy. Nat. Rev. Clin. Oncol. 18, 327–344 (2021).
Wang, Y. et al. First-in-human dose escalation and expansion study of SYSA1801, an antibody-drug conjugate targeting claudin 18.2 in patients with resistant/refractory solid tumors. J. Clin. Oncol. 41, (Suppl. 16), Abstr. 3016 (2023).
ELEVATIONONCOLOGY. First-in-human dose escalation and expansion study of SYSA1801, an antibody-drug conjugate targeting Claudin 18.2 in patients with resistant/refractory solid tumors. https://elevationoncology.com/first-in-human-dose-escalation-and-expansion-study-of-sysa1801-an-antibody-drug-conjugate-targeting-claudin-18-2-in-patients-with-resistant-refractory-solid-tumors/ (2023).
KEYMEDBIO. https://en.keymedbio.com/uploadfile/2021/1217/20211217042321237.pdf (2021).
Jain, N., Smith, S. W., Ghone, S. & Tomczuk, B. Current ADC linker chemistry. Pharm. Res. 32, 3526–3540 (2015).
INNOVENT. Innovent Biologics 2022 Annual Results. https://img.innoventbio.com/Innovent2022AnnualResultsPresentation_MainFile_20230328.pdf (2023).
Qin, S. et al. mRNA-based therapeutics: powerful and versatile tools to combat diseases. Signal Transduct. Target. Ther. 7, 166 (2022).
Sahin, U., Karikó, K. & Türeci, Ö. mRNA-based therapeutics — developing a new class of drugs. Nat. Rev. Drug. Discov. 13, 759–780 (2014).
Bähr-Mahmud, H. et al. Preclinical characterization of an mRNA-encoded anti-claudin 18.2 antibody. Oncoimmunology 12, 2255041 (2023).
Papadopoulos, K. P. et al. A phase I/II dose escalation and expansion trial to evaluate safety and preliminary efficacy of BNT141 in patients with claudin-18.2-positive solid tumors. J. Clin. Oncol. 41 (suppl. 16), TPS2670 (2023).
Weng, Y. et al. The challenge and prospect of mRNA therapeutics landscape. Biotechnol. Adv. 40, 107534 (2020).
Rha, Y. S. et al. Pembrolizumab plus chemotherapy versus placebo plus chemotherapy for HER2-negative advanced gastric cancer (KEYNOTE-859): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 24, 1181–1195 (2023).
Wainberg, Z. A. et al. Bemarituzumab in patients with FGFR2b-selected gastric or gastro-oesophageal junction adenocarcinoma (FIGHT): a randomised, double-blind, placebo-controlled, phase 2 study. Lancet Oncol. 23, 1430–1440 (2022).
Weng, J. et al. Effects of antiemetics on zolbetuximab-induced gastric injury and emesis frequency in ferrets. Ann. Oncol. 34, (Suppl. 2), S198 (2023).
Shitara, K. et al. Management of nausea and vomiting following first-line zolbetuximab + chemotherapy treatment in CLDN18.2+, HER2–, locally advanced unresectable or metastatic gastric or gastroesophageal junction adenocarcinoma: analysis from the phase 3 SPOTLIGHT and GLOW studies. J. Clin. Oncol. 42 (suppl. 3), abstr.372 (2024).
Konecny, G. E. et al. First-in-human phase I study of a novel claudin 6 (CLDN6) targeted antibody drug conjugate (ADC) TORL-1-23. Ann. Oncol. 34, (Suppl. 2), S517 (2023).
Mackensen, A. et al. CLDN6-specific CAR-T cells plus amplifying RNA vaccine in relapsed or refractory solid tumors: the phase 1 BNT211-01 trial. Nat. Med. 29, 2844–2853 (2023).
Türeci, O. et al. A multicentre, phase IIa study of zolbetuximab as a single agent in patients with recurrent or refractory advanced adenocarcinoma of the stomach or lower oesophagus: the MONO study. Ann. Oncol. 30, 1487–1495 (2019).
Acknowledgements
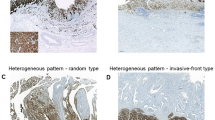
The authors gratefully acknowledge T. Kuwata of National Cancer Center Hospital East, Kashiwa, Japan for providing images used in this publication.
Author information
Authors and Affiliations
Contributions
All authors made a substantial contribution to all aspects of the preparation of this manuscript.
Corresponding authors
Ethics declarations
Competing interests
Y.N. has acted as consultant and/or adviser of Daiichi Sankyo, Exact Sciences Corporation, Gilead Sciences, Guardant Health, Natera, Premo Partners, Roche, Seagen, Takeda Pharmaceutical and has acted as a speaker for Becton Dickinson and Company, CareNet, Chugai Pharmaceutical, Daiichi Sankyo, Eisai, Guardant Health, Genomedia, Hisamitsu, Merck, Miyarisan, MSD, Roche Diagnostics, Seagen, Taiho Pharmaceutical, Tempus Labs, Zeria Pharmaceutical. K.S. has acted as a consultant and/or adviser of ALX Oncology, Inc., Amgen, Astellas, AstraZeneca, Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Daiichi Sankyo, Guardant Health Japan, Janssen, Merck Pharmaceutical, Novartis, Ono Pharmaceutical, Takeda, Zymeworks Biopharmaceuticals and has received (institutional) research funding from Astellas, Amgen, Chugai, Daiichi Sankyo, Eisai, Merck Pharmaceutical, Ono Pharmaceutical, PRA Health Sciences, Syneos Health and Taiho Pharmaceutical. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Clinical Oncology thanks M. Fassan, J. Lim, H. Van Laarhoven and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nakayama, I., Qi, C., Chen, Y. et al. Claudin 18.2 as a novel therapeutic target. Nat Rev Clin Oncol 21, 354–369 (2024). https://doi.org/10.1038/s41571-024-00874-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41571-024-00874-2