Abstract
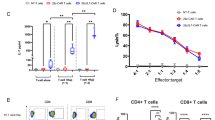
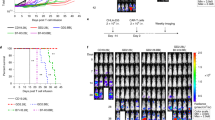
CD19 CAR-T cells have led to durable remissions in patients with refractory B-cell malignancies; nevertheless, most patients eventually relapse in the long term. Many interventions aimed at improving current products have been reported, with a subset of them focusing on a direct or indirect link to the metabolic state of the CAR-T cells. We assessed clinical products from an ongoing clinical trial utilizing CD19-28z CAR-T cells from patients with acute lymphoblastic leukemia. CAR-T clinical products leading to a complete response had significantly higher mitochondrial function (by oxygen consumption rate) irrespective of mitochondrial content. Next, we replaced the carbon source of the media from glucose to galactose to impact cellular metabolism. Galactose-containing media increased mitochondrial activity in CAR-T cells, and improved in in-vitro efficacy, without any consistent phenotypic change in memory profile. Finally, CAR-T cells produced in galactose-based glucose-free media resulted in increased mitochondrial activity. Using an in-vivo model of Nalm6 injected mice, galactose-primed CAR-T cells significantly improved leukemia-free survival compared to standard glucose-cultured CAR-T cells. Our results prove the significance of mitochondrial metabolism on CAR-T cell efficacy and suggest a translational pathway to improve clinical products.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data is available upon request. A pre-print version was posted and can be accessed here: BIORXIV/2023/559091.
References
Kishton RJ, Sukumar M, Restifo NP. Metabolic Regulation of T Cell Longevity and Function in Tumor Immunotherapy. Cell Metab. 2017;26:94–109.
Chang CH, Curtis JD, Maggi LB, Faubert B, Villarino AV, O’Sullivan D, et al. Posttranscriptional control of T cell effector function by aerobic glycolysis. Cell. 2013;153:1239.
Kondo T, Ando M, Nagai N, Tomisato W, Srirat T, Liu B, et al. The NOTCH–FOXM1 axis plays a key role in mitochondrial biogenesis in the induction of human stem cell memory–like CAR-T cells. Cancer Res. 2020;80:471–83.
Vardhana SA, Hwee MA, Berisa M, Wells DK, Yost KE, King B, et al. Impaired mitochondrial oxidative phosphorylation limits the self-renewal of T cells exposed to persistent antigen. Nat Immunol. 2020;21:1022–33.
Labanieh L, Mackall CL. CAR immune cells: design principles, resistance and the next generation. Nature. 2023;614:635–48.
Shah NN, Fry TJ. Mechanisms of resistance to CAR T cell therapy. Nat Rev Clin Oncol 2019;16:372–85.
Cappell KM, Kochenderfer JN. Long-term outcomes following CAR T cell therapy: what we know so far. Nat Rev Clin Oncol. 2023;2023:1–13.
Fraietta JA, Lacey SF, Orlando EJ, Pruteanu-malinici I, Gohil M, Lundh S, et al. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat Med. 2018;24:563–71.
Kawalekar OU, O’Connor RS, Fraietta JA, Guo L, McGettigan SE, Posey AD, et al. Distinct Signaling of Coreceptors Regulates Specific Metabolism Pathways and Impacts Memory Development in CAR T Cells. Immunity. 2016;44:380–90.
Peng J-J, Wang L, Li Z, Ku C-L, Ho P-C. Metabolic challenges and interventions in CAR T cell therapy. Sci Immunol. 2023;8:eabq3016.
Luu M, Riester Z, Baldrich A, Reichardt N, Yuille S, Busetti A, et al. Microbial short-chain fatty acids modulate CD8+ T cell responses and improve adoptive immunotherapy for cancer. Nat Commun. 2021;12:1–12.
Wang T, Gnanaprakasam JNR, Chen X, Kang S, Xu X, Sun H, et al. Inosine is an alternative carbon source for CD8+-T-cell function under glucose restriction. Nat Metab. 2020;2:635–47.
Weinberg F, Hamanaka R, Wheaton WW, Weinberg S, Joseph J, Lopez M, et al. Mitochondrial metabolism and ROS generation are essential for Kras-mediated tumorigenicity. Proc Natl Acad Sci USA. 2010;107:8788–93.
Palaskas NJ, Garcia JD, Shirazi R, Shin DS, Puig-Saus C, Braas D et al. Global alteration of T-lymphocyte metabolism by PD-L1 checkpoint involves a block of de novo nucleoside phosphate synthesis. Cell Discov. 2019;5. https://doi.org/10.1038/s41421-019-0130-x.
Itzhaki O, Jacoby E, Nissani A, Levi M, Nagler A, Kubi A, et al. Head-to-head comparison of in-house produced CD19 CAR-T cell in ALL and NHL patients. J Immunother Cancer. 2020;8:e000148.
Rozenbaum M, Meir A, Aharony Y, Itzhaki O, Schachter J, Bank I, et al. Gamma-Delta CAR-T Cells Show CAR-Directed and Independent Activity Against Leukemia. Front Immunol. 2020;11:1–8.
Jacoby E, Bielorai B, Hutt D, Itzhaki O, Adam E, Bar D, et al. Parameters of long-term response with CD28-based CD19 chimaeric antigen receptor-modified T cells in children and young adults with B-acute lymphoblastic leukaemia. Br J Haematol. 2022;197:475–81.
Sabatino M, Hu J, Sommariva M, Gautam S, Fellowes V, Hocker JD, et al. Generation of clinical-grade CD19-specific CAR-modified CD8 + memory stem cells for the treatment of human B-cell malignancies A platform for the generation of clinical-grade CD19-CAR modified T CD19-CAR modified T mediate superior antitumor responses com. Blood. 2016;128:519–29.
Klein Geltink RI, Edwards-Hicks J, Apostolova P, O’Sullivan D, Sanin DE, Patterson AE, et al. Metabolic conditioning of CD8+ effector T cells for adoptive cell therapy. Nat Metab. 2020;2:703–16.
Sukumar M, Liu J, Ji Y, Subramanian M, Crompton JG, Yu Z, et al. Inhibiting glycolytic metabolism enhances CD8+ T cell memory and antitumor function. J Clin Invest. 2013;123:4479–88.
Wenes M, Jaccard A, Wyss T, Maldonado-Pérez N, Teoh ST, Lepez A, et al. The mitochondrial pyruvate carrier regulates memory T cell differentiation and antitumor function. Cell Metab. 2022;34:731–746.e9.
Bai Z, Woodhouse S, Zhao Z, Arya R, Govek K, Kim D et al. Single-cell antigen-specific landscape of CAR T infusion product identifies determinants of CD19-positive relapse in patients with ALL. Sci Adv. 2022;8. https://doi.org/10.1126/sciadv.abj2820.
Shiratori R, Furuichi K, Yamaguchi M, Miyazaki N, Aoki H, Chibana H, et al. Glycolytic suppression dramatically changes the intracellular metabolic profile of multiple cancer cell lines in a mitochondrial metabolism-dependent manner. Sci Rep. 2019;9:18699.
Aguer C, Gambarotta D, Mailloux RJ, Moffat C, Dent R, McPherson R, et al. Galactose Enhances Oxidative Metabolism and Reveals Mitochondrial Dysfunction in Human Primary Muscle Cells. PLoS One. 2011;6:e28536.
van Bruggen JAC, Martens AWJ, Fraietta JA, Hofland T, Tonino SH, Eldering E, et al. Chronic lymphocytic leukemia cells impair mitochondrial fitness in CD8+ T cells and impede CAR T cell efficacy. Blood. 2019;134:blood.2018885863.
Klysz DD, Fowler C, Malipatlolla M, Stuani L, Freitas KA, Chen Y, et al. Inosine induces stemness features in CAR-T cells and enhances potency. Cancer Cell. 2024;42:266–282.e8.
Weber EW, Parker KR, Sotillo E, Lynn RC, Anbunathan H, Lattin J et al. Transient rest restores functionality in exhausted CAR-T cells through epigenetic remodeling. Science. 2021;372. https://doi.org/10.1126/science.aba1786.
Acknowledgements
The authors thank the production team at the Ella Institute of Immuno-oncology who produced the clinical CAR-T cells. This work was carried out in partial requirements for GG and SA’s MSc degree (Faculty of Medicinal and Health Sciences, Tel Aviv University).
Funding
This work was funded by the Dotan center for hematologic malignancies grant and Israel Cancer Association grant 20240064 (EJ) and NIH grant 5R01CA259635 (TY).
Author information
Authors and Affiliations
Contributions
GG, TY and EJ conceived the initial study plan. GG, SA, AM, YAT, SBY AZ, LS, TY and EJ performed experiments and analyzed the data. AT, TY and EJ supervised the work. GG, TY and EJ wrote the manuscript draft. All authors approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gross, G., Alkadieri, S., Meir, A. et al. Improved CAR-T cell activity associated with increased mitochondrial function primed by galactose. Leukemia (2024). https://doi.org/10.1038/s41375-024-02257-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41375-024-02257-z